Guest writer Kieron Day, Head of Clinical Affairs at Sky Medical Technology, focuses on clinical trials as he talks about commercialisation of medical devices. And this is just one of the challenges faced in bringing innovation to market.

Medical technology (MedTech) device innovation often provides healthcare services with new alternatives to pharmaceutical interventions. Importantly, ultimate adoption of a technology demands that MedTech companies demonstrate clinical evidence that their devices can deliver positive patient outcomes.
Exponential growth in clinical trials
Clinical trials have surged in number in the 21st century. According to a study conducted by ClinicalTrials.gov approximately 100,000 trials in 2010 grew to more than 390,000 in 2021. This undoubtedly reflects the growing need for new medical solutions, coupled with heightened regulatory requirements. Medical technology adoption is essential because it can ease the pressure on healthcare systems. However for a device to be allowed onto the market and for clinicians to be convinced of its merits, companies require evidence; proving that a technology improves patient outcomes for a rapidly ageing population is imperative.
The sheer volume of clinical trials hints at a vast and ongoing unmet clinical need. After all the clinical trial is the starting point from which MedTech can help global healthcare systems. The goal is for today’s clinical trials to ultimately solve issues associated with rising costs and growing demand. Not only does healthcare innovation benefit overwhelmed medical professionals, but it enhances patient outcomes. Fewer complications and faster recovery translates into efficient healthcare delivery and lower cost.
The number of clinical trials continues to climb. This indicates huge commercial competition in modern healthcare. Moreover, the demand for definitive evidence means the task of creating and executing large scale, multi-centre clinical trials will become more challenging.
Exploring the full potential
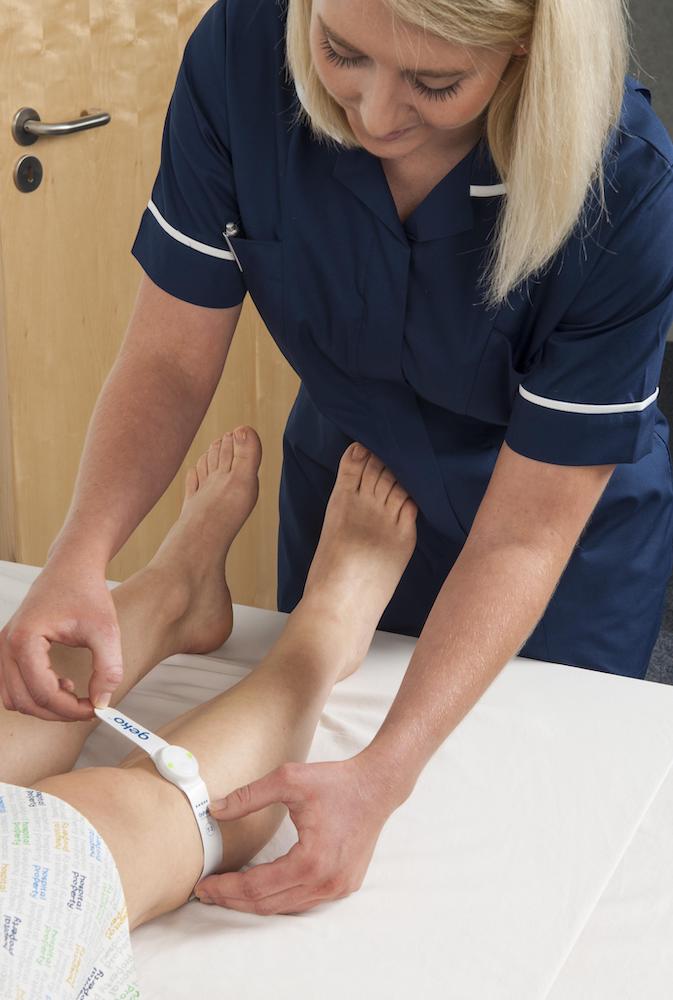
Executing successful clinical trials is always complicated. This is particularly so when there are various medical conditions a device could potentially treat. At Sky Medical Technology, we have developed the wearable geko™ device that is clinically proven to promote blood flow. This means, in theory its potential therapeutic applications could include any condition that can be cured or improved with increased circulation. As a result, reduced swelling, faster healing wounds, and the prevention of deep vein thrombosis (DVT) are within its gift. Devices with broad application are recognised as ‘platform technologies’.
While platform technology devices can provide a plethora of benefits for healthcare systems, this does not mean that clinical evidence in one area is applicable in others. geko™ is a good example. At Sky we have already completed the appropriate certification to use the geko™ device on patients, demonstrating that it can improve blood flow significantly. We then still need to also prove its effectiveness in addressing postoperative oedema in a proof-of-concept trial. We must generate specific evidence to ensure devices are viable across each new medical application.
Difficult conditions for clinical studies
The COVID-19 crisis created a difficult environment for completing successful clinical trials. Indeed it brought many outpatient clinics to a standstill. When it came to clinical trials, multiple centres actively recruiting patients closed during lockdown. Medical resources have been shifted, and healthcare systems have been overwhelmed. This has been disastrous for medical services that are classed as non-essential. Strikingly, many patients have either not had the treatments they need or have developed other illnesses during the pandemic.
Accessing clinical support
All clinical trials rely on the goodwill of key opinion formers within the clinical environment. These are busy people, even more so in a post-COVID environment. It is important to win the confidence of well-published clinicians, so it is worth carefully considering how best to introduce the technology itself. The clinician may then further evaluate a device.
Medical technology adoption relies on developing trust and sustainable relationships with clinicians. Moreover the ideal clinician partner is also well connected globally. In essence, solid relationships in the UK can facilitate introduction to clinician peers in the US.
Invest, invest, invest
Rules and regulations around MedTech adoption are becoming increasingly strict. Devices need to comply with clinical regulation as well as regulation around electrical safety and biocompatibility. This requires significant investment into clinical trials, both in terms of time and money.
Typically, companies will pay for trials, write and manage the protocol and verify the data. These so-called ‘industry-sponsored studies’ can sometimes be grant-funded. Ultimate responsibility for the safety of the patient falls on the company’s shoulders.
In contrast, investigator-sponsored studies see a clinician wanting to investigate use the device for a specific purpose. In such cases, the physician owns, analyses, and publishes the data. This can clearly benefit MedTech companies which may contribute towards the cost of the study. Investigator-sponsored studies can also help companies build credibility and a good reputation within the industry.
Paving the way to commercial success
Patience is paramount when building an evidence base to support commercial deployment of MedTech. The regulatory pathway is at least three years long. Then there is a further three to five years to create commercial and marketable data. It is not viable to do all the trials immediately, across every medical condition and every country.
Proving a device is safe is the first of many steps during the lifetime of a medical technology. Companies need to prove that they deliver positive patient outcomes and provide new data for each of the device’s uses. And once the pre-market requirements are met, post-marketing surveillance of all aspects of its performance remain an ongoing responsibility. MedTech companies should view investment in the clinical trial process as an investment in their own future. It will pay significant dividends in terms of commercial success.
Source: Kieron Day, Sky Medical Technologies
Editor’s note: At Medlatest we occasionally enjoy contributions from guest writers. If you would like to submit a relevant editorial for our consideration, contact us here.
published: December 1, 2021 in: medlatest Editorial, News, Technology

