Oxford, UK company, IMed Consultancy has launched a free whitepaper on the theme of Post Marketing Surveillance. The company says … continue reading “Post Market Surveillance: Beyond Pain to Unlocking Potential- New Free Whitepaper by IMed Consultancy”
Regulatory
Valvosoft Non-Invasive Aortic Stenosis Therapy: Pivotal Study Results Promising
Cardiawave SA is the developer of the Valvosoft Non-Invasive Ultrasound Therapy (NIUT) device for the treatment of severe symptomatic calcific … continue reading “Valvosoft Non-Invasive Aortic Stenosis Therapy: Pivotal Study Results Promising”
Thrombectomy Specialist Penumbra’s Lightning Products CE Mark Makes Them Europe’s Only CAVT Systems
Thrombectomy technology leader, Penumbra Inc. has secured CE Mark in Europe for the latest computer assisted vacuum thrombectomy (CAVT) devices. Background … continue reading “Thrombectomy Specialist Penumbra’s Lightning Products CE Mark Makes Them Europe’s Only CAVT Systems “
ACURATE Prime™, Boston Scientific’s New Aortic Valve System Gains CE Mark
Boston Scientific Corporation tells us it has obtained CE mark for the ACURATE Prime™ Aortic Valve System. Indeed it is … continue reading “ACURATE Prime™, Boston Scientific’s New Aortic Valve System Gains CE Mark”
Emboliner ® IDE Study 100 Patient Milestone
The Emboliner “Protect the Head to Head” investigational device exemption (IDE) clinical trial has seen its one hundredth patient enrolled. … continue reading “Emboliner ® IDE Study 100 Patient Milestone”
VARIPULSE™ Pulse Field Ablation (PFA) Platform Gains CE Mark
Biosense Webster, Inc., has announced European CE mark approval of the VARIPULSE™ Platform. The approval covers the treatment of symptomatic drug refractory … continue reading “VARIPULSE™ Pulse Field Ablation (PFA) Platform Gains CE Mark”
Foldax Signs Indian Manufacturer to Help Expedite Mitral Valve Commercialization in the Country
Heart Valve Pioneer Foldax® Inc., will see its innovative TRIA™ polymer mitral surgical heart valves being manufactured in India. A newly … continue reading “Foldax Signs Indian Manufacturer to Help Expedite Mitral Valve Commercialization in the Country”
Medtech Regulatory and Commercial Landscape Considerations: UK, US, or EU first?
Feature article by Timothy Bubb, Medtech regulatory specialist and TechnicalDirector at IMed consultancy. Technology is taking an increasingly central place … continue reading “Medtech Regulatory and Commercial Landscape Considerations: UK, US, or EU first?”
Scoliosis Management Boost as FDA Clears AI-Solution
Scoliosis management has gained a new string to its bow in the United States. The FDA has cleared NSite Medical’s … continue reading “Scoliosis Management Boost as FDA Clears AI-Solution”
Spyral ™ Gains FDA Approval
Medtronic plc says the US FDA has approved the Symplicity Spyral ™ renal denervation (RDN) system as an adjunct to … continue reading “Spyral ™ Gains FDA Approval”
FDA approves Recor Medical’s Paradise™ Ultrasound Renal Denervation for Hypertension
Recor Medical, Inc. (“Recor”) and its parent company, Otsuka Medical Devices Co., Ltd. (“Otsuka Medical Devices”) have announced the U.S. … continue reading “FDA approves Recor Medical’s Paradise™ Ultrasound Renal Denervation for Hypertension”
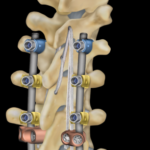
FDA Clears First Spine Device for Ligament Augmentation
Medtronic plc, has received U.S. FDA 510(k) clearance and Breakthrough Device designation for its novel LigaPASS™ 2.0 Ligament Augmentation System. … continue reading “FDA Clears First Spine Device for Ligament Augmentation”
First Limus Drug-eluting Balloon for US as SELUTION SLR™ Gets FDA IDE Approval
MedAlliance’s SELUTION SLR™ drug-eluting balloon (DEB) has received FDA Investigational Device Exemption (IDE) approval, making it the first limus DEB … continue reading “First Limus Drug-eluting Balloon for US as SELUTION SLR™ Gets FDA IDE Approval”
CVRx®Barostim™ Heart Failure System Gains MR-Conditional Labeling Approval
CVRx, Inc., developer of the world’s first FDA-approved neuromodulation device to treat the symptoms of heart failure, has received U.S. … continue reading “CVRx®Barostim™ Heart Failure System Gains MR-Conditional Labeling Approval”
Japanese PMDA Approval for BioFreedom™ Ultra and US FDA Approval for BioFreedom™
Biosensors International Group, Ltd. has announced the Japanese PMDA Approval for BioFreedom™ Ultra and US FDA Approval for BioFreedom™. Background … continue reading “Japanese PMDA Approval for BioFreedom™ Ultra and US FDA Approval for BioFreedom™”
First O.U.S. Patients Get TRIA Biopolymer Surgical Aortic Valve™
Foldax®, Inc. says the first patients have been enrolled in the Drugs Controller General of India (DCGI)-approved clinical trial of … continue reading “First O.U.S. Patients Get TRIA Biopolymer Surgical Aortic Valve™”