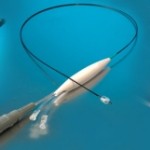
Thrombectomy technology leader, Penumbra Inc. has secured CE Mark in Europe for the latest computer assisted vacuum thrombectomy (CAVT) devices. Background … continue reading “Thrombectomy Specialist Penumbra’s Lightning Products CE Mark Makes Them Europe’s Only CAVT Systems “
Vascular
Jupiter Endovascular Funding Means Green Light for Pulmonary Embolism Pivotal Trial
New funding will be used to support Jupiter Endovascular in its upcoming pivotal trial for Pulmonary Embolism. It will also … continue reading “Jupiter Endovascular Funding Means Green Light for Pulmonary Embolism Pivotal Trial”
TCAR Count at 100k for Silk Road Medical
Stroke Device specialist Silk Road Medical, Inc. tells us that more than 100,000 TCAR procedures have been performed to date. … continue reading “TCAR Count at 100k for Silk Road Medical”
Hemodialysis Access Promise from Polymer Implant
Netherlands and US-based medtech company Xeltis tells us about its first-in-human (FIH) vascular conduit trial for hemodialysis access. Vascular surgeon … continue reading “Hemodialysis Access Promise from Polymer Implant”
Spyral ™ Gains FDA Approval
Medtronic plc says the US FDA has approved the Symplicity Spyral ™ renal denervation (RDN) system as an adjunct to … continue reading “Spyral ™ Gains FDA Approval”
Venous Leg Ulcer Treatment Options Expanded with UK Reimbursement Grant
UK-based medical device manufacturer Sky Medical Technology Ltd tells us its wearable geko device is now reimbursed in the UK. This means better access … continue reading “Venous Leg Ulcer Treatment Options Expanded with UK Reimbursement Grant”
FDA approves Recor Medical’s Paradise™ Ultrasound Renal Denervation for Hypertension
Recor Medical, Inc. (“Recor”) and its parent company, Otsuka Medical Devices Co., Ltd. (“Otsuka Medical Devices”) have announced the U.S. … continue reading “FDA approves Recor Medical’s Paradise™ Ultrasound Renal Denervation for Hypertension”
SELUTION SLR Coronary Sirolimus DEB Study: First US Patient Enrolled
The first US patient has been enrolled at Medstar Washington Hospital Center in the SELUTION4ISR study evaluating SELUTION SLR™ to … continue reading “SELUTION SLR Coronary Sirolimus DEB Study: First US Patient Enrolled”
New Commercial Operations VP for LimFlow
LimFlow SA has announced the appointment of Mike Mathias as Vice President of Commercial Operations. The company is a pioneer in … continue reading “New Commercial Operations VP for LimFlow”
Venous Leg Ulcers; Economics and MedTech’s Answers
Technological advancements such as Sky Medical Technology‘s wearable geko™ device mark a new era in the treatment of Venous Leg … continue reading “Venous Leg Ulcers; Economics and MedTech’s Answers”
First Limus Drug-eluting Balloon for US as SELUTION SLR™ Gets FDA IDE Approval
MedAlliance’s SELUTION SLR™ drug-eluting balloon (DEB) has received FDA Investigational Device Exemption (IDE) approval, making it the first limus DEB … continue reading “First Limus Drug-eluting Balloon for US as SELUTION SLR™ Gets FDA IDE Approval”
$40M New Funding to Prevent Amputations
LimFlow SA, has closed a $40 million (€36 million) oversubscribed Series D financing round. The company is a pioneer in the … continue reading “$40M New Funding to Prevent Amputations”
LimFlow Completes Enrollment in PROMISE II Pivotal Trial
LimFlow SA tells us that it has completed patient enrollment in the pivotal trial of the LimFlow System for chronic limb-threatening … continue reading “LimFlow Completes Enrollment in PROMISE II Pivotal Trial”
Continuous Aspiration Thrombectomy with Penumbra Indigo® System CAT™RX Safe and Effective Says New Study
Success for Penumbra Inc.’s Indigo® System CATTM RX Catheter in a newly reported study. The device met the primary endpoint and demonstrated high rates of blood clot … continue reading “Continuous Aspiration Thrombectomy with Penumbra Indigo® System CAT™RX Safe and Effective Says New Study”
Renal Denervation Back in Europe with ReCor Paradise™ Launch
Treatment supported by evidence from multiple studies and Renal Denervation Position Paper from the European Society of Hypertension Background It’s … continue reading “Renal Denervation Back in Europe with ReCor Paradise™ Launch”
FDA Breakthrough Device Status for Hancock Jaffe VenoValve®
Cardiac and Vascular device developer Hancock Jaffe Laboratories, Inc., tells us that the U.S. FDA has granted Breakthrough Device Designation status to … continue reading “FDA Breakthrough Device Status for Hancock Jaffe VenoValve®”