In short
Sunshine Heart, Inc. has featured on our pages recently with the news that its C-Pulse ® Heart Assist System was CE marked in July and then that the FDA was giving it the glad eye in August. It’s not all that surprising then that the company has now announced that it has received conditional approval from the FDA for an Investigational Device Exemption (IDE) for its flagship device.
Background
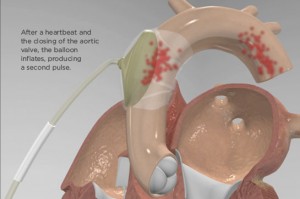
The C-Pulse Heart Assist System, or C-Pulse System utilises the scientific principles of intra-aortic balloon counter-pulsation applied in an extra-aortic approach to assist the left ventricle by reducing the workload required to pump blood throughout the body, while increasing blood flow to the coronary arteries. Operating outside the patient’s bloodstream, the extra-aortic approach of the C-Pulse technology offers greater flexibility, allowing patients to safely disconnect to have intervals of freedom to perform certain activities such as showering.
The C-Pulse System may help maintain the patient’s current condition and, in some cases, reverse the heart failure process, thereby potentially preventing the need for later stage heart failure therapies, such as left ventricular assist devices (LVADs), artificial hearts or transplants.
With the receipt of conditional approval of the IDE, Sunshine Heart plans to initiate a pivotal trial in North America in the fourth quarter of 2012. The trial design will consist of 388-patients of which half will be implanted with the C-Pulse System. The other half will be randomized to optimal medical therapy across 30-40 clinical sites. Sunshine Heart expects to receive revenues from trial sites for device implants as the FDA has granted CMS Category B3 status. Because of this designation, it is also anticipated that participating trial centers will be reimbursed by CMS and most private insurance providers.
“Having led the C-Pulse feasibility study last year and witnessed the potential impact the device had in patients suffering from heart failure, I am excited to return to play an integral role in C-Pulse’s final stages of development,” said Dr. Abraham. “With a large and growing population of C-Pulse eligible patients and hospitals facing financial penalties for high heart failure rehospitalisation rates beginning in 2013 under the Obama Health Reform Act, the C-Pulse pivotal trial will determine if it will be beneficial to both patients and hospitals alike to improve health outcomes.”
“We are thrilled to receive conditional approval from the FDA to move forward into a pivotal trial for C-Pulse,” said Dave Rosa, CEO of Sunshine Heart. “Today’s announcement represents another significant milestone for the Company, as well as for the C-Pulse System. I am especially pleased that we were able to receive this approval on our initial submission. The Company has achieved its regulatory and clinical objectives by receiving both the CE Mark and U.S. IDE approval this year. These timely approvals continue to support the Company’s market guidance and timelines.”
Source: Sunshine Heart Inc.
published: October 2, 2012 in: Approval/Clearance, Cardio, Clinical Studies/Trials
