In short
Californian vascular device company Endologix, Inc., has gained CE Mark approval of the current version of its unique Nellix® EndoVascular Aneurysm Sealing System which involves filling the aneurysm sac with a bag full of a biostable polymer in patients with abdominal aortic aneurysms (“AAA”). The device is undergoing enhancements prior to a planned launch next year.
Background
Endologix, Inc. develops and manufactures minimally invasive treatments for aortic disorders. We reported on thie EU efforts earlier this year, here. But in case you’ve not come across them, they’re based in Irvine, California and will sell over a hundred million dollars worth of aorta-focused devices this year, mainly endovascular stents, more than 80% of revenue coming from the U.S. With its developing presence in Europe you can expect the company’s presence to become increasingly obvious on the EU side of the pond too.
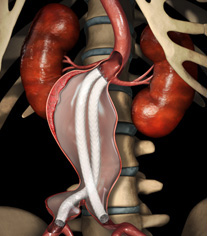
Nellix is an intriguing and innovative new endovascular aneurysm sealing (“EVAS”) system designed to simplify endovascular procedures, treat a broader range of patients, and provide enhanced clinical outcomes. Fundamentally the device works by inserting what is descriptively called an “endobag” full of a proprietary biostable polymer into the aneurysm sac, having first inserted twin endovascular grafts to maintain blood flow. This “space filler” acts to generate a seal around the grafts and from clinical data seems to work, most notably providing a treatment option for patients who may not be suitable for other techniques.
So when will you see the product? Well, Endologix says it is currently implementing a few enhancements to the Nellix System intended to further optimise the device for commercialisation. When complete, the Company will submit these enhancements to its Notified Body, with expectations of gaining CE Mark approval for the commercial version of the device and beginning a limited market introduction of the enhanced system in Europe by the end of the second quarter 2013.
Company comments
John McDermott, President and Chief Executive Officer, said, “Nellix has the potential to treat more AAA patients and get better clinical outcomes than any other device for the endovascular repair of AAA. Receiving CE Mark for the current version of the system is an important regulatory milestone and we look forward to providing this ground-breaking technology to physicians and their patients in 2013.”
Regulatory status
The Nellix System is not approved in the United States for either investigational use or commercial sale.
Source: Endologix, Inc., Globe Newswire
published: September 7, 2012 in: Approval/Clearance, Vascular
