 In short
In short
Renal denervation for treatment of hypertension is pretty of-the-moment, as we’ve reported in recent months. Only last week we covered reports of sustained reduction in hypertension in a study using Medtronic’s Symplicity™ system, which can be found here, but we’ve also covered the offerings from St.Jude and German company Recor Medical’s ultrasound-based approach. Now another company, Vessix Vascular Inc., has announced the CE marking of its own percutaneous radiofrequency (RF) balloon catheter technology. The company’s V2 Renal Denervation System™ uses RF energy to disrupt renal sympathetic nerves whose hyperactivity leads to uncontrolled high blood pressure.
Background
Hypertension is the leading attributable cause of death worldwide. According to the American Heart Association, a 5 mm Hg (millimeters of mercury) reduction in systolic blood pressure results in a 14 percent decrease in stroke, a 9 percent decrease in heart disease and a 7 percent decrease in overall mortality. Renal denervation has shown in published clinical studies to be safe, durable and effective in reducing systolic blood pressure by approximately 20 percent.
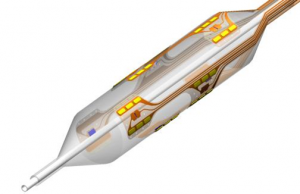
The V2 System for the treatment of uncontrolled hypertension is an over-the-wire balloon catheter with an array of RF electrodes mounted in a precise pattern designed to deliver a modest dose of RF energy to disrupt the renal nerves located in the adventitia that surround the renal artery. The patented RF balloon catheter connects to a proprietary bipolar RF generator specifically designed and optimised for the renal denervation clinical application.
Therapy is delivered via a balloon catheter that is quite familiar to interventional cardiologists and other physicians, thereby increasing the usability and safety profile of the device.
The V2 balloon catheter occludes blood flow to the renal artery during the 30-second RF therapy delivery allowing for a directed and highly controlled flow of heat energy to the target nerves. Consequently, Vessix claims its V2 is significantly faster – by an order of magnitude – than other renal denervation systems currently marketed.
Rapid RF treatment time greatly increases the efficiency of the denervation procedure and additional safety benefits to the clinician conducting the procedure and the patient in terms of less use of contrast dye and lower exposure to radiation.
Company comments
“Vessix offers a unique approach to treating uncontrolled hypertension that provides excellent clinical results and is faster, easier to use and less painful for patients than any renal denervation system currently available in the market,” said CEO Raymond W. Cohen. “After eight years optimizing RF balloon catheter and bipolar RF generator technologies, it is rewarding to see the V2 System working effectively in clinical practice to safely reduce patient blood pressures. As part of our commercialization strategy, Vessix has initiated a post-market approval surveillance study during which we plan to treat 120 patients at up to 20 international centers located across Western Europe.”
Michael D. Gioffredi, Vessix’s Chief Commercialization Officer said, “Renal denervation is rapidly becoming one of the most important medical device developments in decades and we believe that our unique design will set a new standard for the treatment. In light of this regulatory clearance and the strong early clinical results from our multi-center European pilot study, we are moving forward with our commercialization plans in Europe and we look forward to sharing additional pre-clinical and human data at upcoming medical conferences in support of that launch.”
SOURCE Vessix Vascular, Inc., PR Newswire
published: May 2, 2012 in: Approval/Clearance, Clinical Studies/Trials, Medtronic, St Jude, Vascular
