In short
St. Jude Medical, Inc. has announced it has received European CE Mark approval of its Eon™ family of neurostimulators for patients with intractable chronic migraine.
Unveiled at the European Headache and Migraine Trust International Congress in London, this approval includes the Eon Mini™ neurostimulator, which is the world’s smallest rechargeable neurostimulator with the longest-lasting battery in its class, and the Eon and EonC™ neurostimulators.
Preceding the Eon family approvals, a year ago the company received European CE Mark for its Genesis™ neurostimulation system, the industry’s first regulatory approval for an implanted neurostimulation device to treat patients with intractable chronic migraine. We covered that story at the time, here.
Background
Estimates by the World Health Organization (WHO) indicate that 10 percent of adults worldwide suffer from migraine and 1.7 to 4 percent of adults have headaches 15 or more days per month. It is claimed that migraine ranks as one of the top 20 most disabling conditions in the world, according to WHO and in terms of monetary cost, estimates say it is one of the most expensive neurological disorders. According to the European Journal of Neurology the total annual cost attributed to migraine amounts to €111 billion in the EU alone.
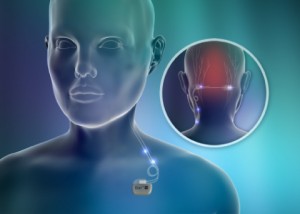
St.Jude’s Eon and Genesis systems deliver peripheral nerve stimulation (PNS) of the occipital nerves to manage the pain and disability associated with intractable chronic migraine. This type of migraine is defined as headache lasting at least four hours per day for 15 or more days per month, causing at least moderate disability, and not responding to three or more preventive drugs. PNS therapy for this condition involves the delivery of mild electrical pulses to the occipital nerves that are located just beneath the skin in the back of the head. A small electrical lead or leads are placed under the skin and connected to the neurostimulator, which produces the pulses of stimulation.
Prior to receiving these approvals, St. Jude Medical conducted a large scale double-blind, randomised, controlled clinical study evaluating PNS to treat the pain and disability associated with chronic migraine. After 12 weeks of stimulation, patients reported an average of six fewer headache days a month. After one year of stimulation, 65 percent of patients reported excellent or good pain relief and 89 percent said they would recommend the procedure to someone else. Study data were presented at the International Headache Congress in 2011 and have been accepted for publication.
Physician comments
“Intractable chronic migraine is one of the most difficult-to-treat headache disorders,” said Professor Gennaro Bussone, M.D., head of the Neurological Department at Istituto Besta in Milan Italy. “By definition, people living with this condition are spending half their month living with debilitating headaches. This therapy expands our options in helping manage patients who suffer with disabling chronic migraine symptoms.”
Company comments
“Neurostimulation technology represents an exciting new approach to treat intractable chronic migraine,” said Eric S. Fain, M.D., president of the St. Jude Medical Implantable Electronic Systems Division. “We are proud to be able to offer this potentially life-changing therapy for patients who suffer with this debilitating condition and so desperately need a more effective treatment option.”
The products
Eon Mini Neurostimulator
Approximately the size of a man’s watch, the rechargeable Eon Mini neurostimulator has a thin 10-mm profile and weighs 29 grams (approximately 1.0 oz). Its small size allows for a smaller incision, which gives physicians increased flexibility in selecting the implant location and is intended to make the site less visible and more comfortable for patients.
Eon Neurostimulator
Featuring a rechargeable battery, the Eon neurostimulator is designed to provide stimulation over a long period of time, making it a good choice for patients who require high-power stimulation settings and need to use the system for a large percentage of the day. The Eon device can provide sustainable therapy and maintain a reasonable recharge interval for 10 years, potentially resulting in fewer battery replacement surgeries.
EonC Neurostimulator
The EonC neurostimulator provides a convenient option for patients who prefer or require the simplicity of a non-rechargeable medical device. The device features the greatest battery capacity of any primary cell neurostimulator.
St. Jude Medical’s neurostimulators are the only fully implantable neurostimulation devices approved in Europe for the management of the pain and disability associated with intractable chronic migraine. They are also approved in many countries for the management of chronic pain of the trunk and limbs.
Source: St.Jude Medical, Inc., Business Wire
published: September 24, 2012 in: Approval/Clearance, Neuro, St Jude
