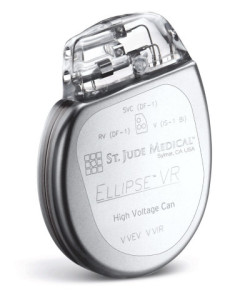
An expanded CE Mark approval for St. Jude Medical’s Ellipse™ implantable cardioverter defibrillator (ICD) means existing or future patients can now undergo magnetic resonance imaging (MRI) scans without compromising device performance. The newly expanded approval also applies to the company’s Durata™ and Optisure™ defibrillation leads.
Background
Designed to offer patients added protection in the event of a life-threatening arrhythmia, the Ellipse device is among the smallest and most advanced ICDs today. The device uses St. Jude Medical’s DynamicTx™ algorithm, which automatically adjusts shocking configurations for patients in order to ensure the delivery of high-voltage therapy if an electrical short occurs in one part of the system.
The Ellipse ICD also utilizes something called DeFT Response™ technology, allowing for physicians to customize the amount of energy delivered for each individual patient. Furthermore, it features a low friction coating which has been demonstrated in testing to significantly reduce the friction between the device and leads, potentially reducing lead-to-can abrasion, one of the most common types of insulation failure in the industry.
St. Jude Medical says it anticipates that the coming months will see updated labeling for many of its existing cardiac rhythm management devices, which will allow more patients the ability to safely undergo MRI scans. Indeed the company is planning to submit test data in key markets around the world in 2015 for MRI conditional labeling on additional existing high-voltage products including the Fortify™ Assura ICD, Quadra Assura CRT-D, and Quartet LV leads.
St. Jude has also announced that it has received CE Mark approval for its Assurity MRI™ and Endurity MRI™ pacemaker device families. These lay claim to being the world’s smallest, longest-lasting wireless pacemakers available to patients today and allow early notification of atrial fibrillation-related events and pacing that helps reduce heart failure-related hospitalizations.
Company comments
“St. Jude Medical is committed to providing MRI-compatible solutions for its ICD and pacemaker devices for patients and physicians worldwide,” said Eric S. Fain, M.D., group president of St. Jude Medical. “These products are already proven cardiac rhythm management technologies and now, due to extensive research and testing, they are available in the MRI environment. We will continue to develop advanced MRI-compatible technologies in order to ensure patient access to this important diagnostic tool.”
Physician comments
“It’s not uncommon for patients with cardiac devices to have an unrelated medical need for an MRI scan over the lifetime of their device,” said Amir Zaidi, M.D., consultant cardiologist at the Manchester Royal Infirmary in Manchester, U.K. “The growing availability of MRI-compatible technologies offered by St. Jude Medical is significant for patients, because it now means they will have access to this important diagnostic tool without compromising their device performance.”
Source: Business Wire
published: May 14, 2015 in: Approval/Clearance, Cardio, Company News, St Jude