 In short
In short
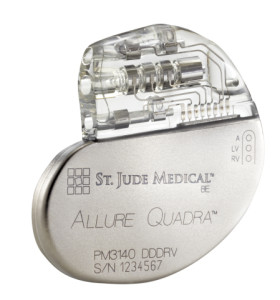
St. Jude Medical, Inc. has announced CE Mark approval and European launch of its Allure Quadra™ Cardiac Resynchronization Therapy Pacemaker (CRT-P). The device brings quadripolar lead technology to the pacemaker market for the first time.
Background
Approximately one in 10 patients who receive a bipolar CRT system has complications that could require additional surgery. St.Jude Medical’s quadripolar technology allows for additional pacing configurations that provide physicians with options not available on traditional bipolar systems. These additional options help manage common pacing complications without exposing the patient to additional surgeries for lead repositioning.
St. Jude says this CRT-P system offers more pacing options to deliver higher success rates, citing the existence of robust clinical data. Quadripolar leads allow for increased implant efficiencies, which clinical data indicates can result in fewer surgical revisions. The Allure family of devices also offer enhanced heart failure (HF) diagnostics, including CorVue™ Impedance Monitoring, for improved patient management.
Additionally, the Allure CRT-P device improves HF management with timely access to vital diagnostic data including CorVue™ Impedance Monitoring for earlier insight into HF progression, and Direct Trend™ Reports for a simplified look at all device data. The Allure Quadra CRT-P integrates the best-in-class quadripolar technology with multiple pacing configurations and TailoredTherapy™ features designed to enable physicians to address the dynamic challenges of HF by optimizing the quadripolar system at implant and follow-up.
Physician comments
“Adding quadripolar technology to the CRT-P platform moves us forward in the treatment of heart failure and allows us to expand the proven clinical benefits to a new patient population,” said Dr. Amir Zaidi, consultant cardiologist and devices lead at Manchester Royal Infirmary, England. “The new features and additional pacing options allow for optimal lead placement and provide the patient greater opportunities to respond to therapy, while reducing the need for re-intervention.”
Company comments
“Since its launch in 2010, the St. Jude Medical quadripolar CRT-D platform has allowed physicians to more effectively treat their patients and we expect the same results with our quadripolar CRT-P line,” said Eric S. Fain, M.D., president of the Implantable Electronic Systems Division at St. Jude Medical. “We believe these benefits result in a cost-effective solution that elevates the standard of care for heart failure patients in need of cardiac resynchronization therapy.”
Regulatory status
The Allure Quadra CRT-P device is currently not approved for use in the U.S.
Source: St.Jude Medical, Inc., Business Wire
published: April 30, 2013 in: Approval/Clearance, Cardio, St Jude
