In short
Boston Scientific Corporation has closed its acquisition of Californian Cardiac technology company Cameron Health, Inc., and, as a result, added to its product portfolio the world’s first and only commercially available subcutaneous implantable cardioverter defibrillator, called the S-ICD® System.
Background
We first covered this story back in April, here, when news of Bsoton’s acquisition of Cameron Health was first announced. Since then we’ve returned to the tale on a couple of occasions as the FDA panel met to review the company’s leadless implantable cardioverter defibrillator. Most recently we covered the favourable outcome of the FDA panel vote, which is seen as a precursor to full FDA approval, anticipated to be granted in the next 12 months.
In a press release Boston Scientific describes the acquisition as “the capstone of a nearly 10-year relationship between the two companies during which Boston Scientific invested in Cameron Health during its ground-breaking research and product commercialisation efforts.
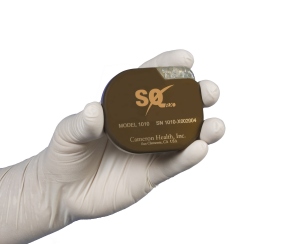
Developed by Cameron Health, the entire S-ICD System sits just below the skin, leaving the heart and blood vessels untouched, offering patients an alternative to conventional transvenous ICDs, which require leads to be placed into the heart itself. In that sense it offers a new class of protection against sudden cardiac arrest.
Transaction Details
The transaction follows Boston Scientific’s exercise of its option to acquire Cameron Health announced on March 8, 2012. Under the terms of the agreement, Boston Scientific paid $150 million at closing. The agreement calls for an additional potential payment of $150 million to be made upon FDA approval of the S-ICD System and up to an additional $1.050 billion of potential payments to be made upon the achievement of specified revenue-based criteria over a six-year period following FDA approval.
Company comments
for Boston Scientific
“We are pleased to complete the acquisition of Cameron Health, furthering Boston Scientific’s commitment to introducing innovation in the CRM space,” said Hank Kucheman, chief executive officer at Boston Scientific. “Boston Scientific now provides physicians and their patients with an option to choose either the industry’s thinnest, longest-lasting transvenous ICD or the world’s first and only commercially available completely subcutaneous ICD.”
for Cameron Health
“We believe that the combination of Cameron Health’s breakthrough technology and Boston Scientific’s already strong arrhythmia management product portfolio and commercial capabilities will help unlock the enormous potential of the S-ICD System,” said Kevin Hykes, former chief executive officer of Cameron Health, who will continue to lead the S-ICD team at Boston Scientific. “Equally exciting is the promise of next-generation subcutaneous technology that we expect will continue to expand the reach of ICD therapy to more patients.”
Regulatory status
The S-ICD System received CE Mark in 2009 and is commercially available in many countries in Europe, as well as New Zealand. To date, more than 1,300 devices have been implanted in patients around the world.
The S-ICD® System is an investigational device and limited under U.S. law to investigational use only, and is not available for sale in the U.S.
Source: Boston Scientific Corporation, PR Newswire
published: June 11, 2012 in: Boston Scientific, Cardio, Mergers and Acquisitions, News
