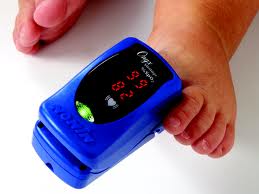
In short
It’s been the Thoracic specialties this week with the holding of the ATS meeting in San Francisco’s Moscone Centre. Minnesota based Nonin Medical, Inc., the inventor of finger pulse oximetry and a leader in noninvasive medical monitoring, used the event to announce commercial release with expanded labelling claims for its Onyx® Vantage 9590 professional fingertip pulse oximeter. The new labelling, cleared under FDA 510(k) regulations, allows the Onyx Vantage to be used on thumbs and toes – an addition to a previous indication for fingertips – on paediatric through adult patients whose digit diameters range from 0.3 inches to 1.0 inches.
Background
The Onyx Vantage finger pulse oximeter is engineered with Nonin Medical’s clinically proven PureSAT™ pulse oximetry technology – the same accurate technology used in its full line of oximeters.
The device accurately captures oxygen saturation (SpO2) and pulse rate measurements even in the presence of low perfusion and dark skin pigmentation. It is the first and only fingertip pulse oximeter with accuracy claims for expanded use on thumbs and toes.
Company comments
“Nonin’s Onyx brand of professional finger pulse oximeters is already proven accurate in the widest range of patients and settings,” said Kevin McGowan, vice president, global sales, for Nonin Medical. “With this expanded indication, clinicians will now be able to use Nonin’s newest professional pulse oximeter, the Onyx Vantage 9590, on fingers, thumbs or toes. This gives them even more flexibility to obtain accurate readings on a wide range of patients from pediatrics through adults.
“No longer will clinicians have to trade flexibility for peace of mind,” McGowan added. “That’s because, regardless of which digit they use, they can be assured that they are getting accurate readings for improved decision making that may lead to earlier intervention and better outcomes.”
Source: Nonin Medical Inc.
published: May 23, 2012 in: Alerts/Adverse Events, Launches / Withdrawals, News, Thoracic/Respiratory
