In short
Last December we announced that Texas-based medical device manufacturer BiO2 Medical, Inc., had gained CE Mark approval for its triple lumen Angel™ Catheter. Now, just to keep you in the picture the company, has reportedly sold its first unit in the European Union.
Background
The Angel Catheter is used to prevent pulmonary embolism in critically ill patients, remembering that between 200,000 and 300,000 people die annually from pulmonary embolism in the United States alone.
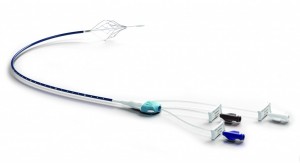
The catheter comprises a Nitinol, Inferior Vena Cava (IVC) filter, permanently attached to a central venous catheter (CVC) for use in preventing Pulmonary Embolism (PE) in critically ill patients. Placed at the patient’s bedside without the need for fluoroscopy, the Angel™ Catheter provides instant protection from PE and is the first IVC Filter to receive CE Mark approval for prophylactic indications.
The design of the Angel™ Catheter allows for easy insertion and access to the central venous system for administering medications, fluids, or blood products; blood sampling; and monitoring of central venous pressure. It is intended to be used during the critical time period in which anticoagulation therapy poses a high risk of complications, including major bleeding and death, and in which patients are at highest risk of VTE. The filter can be easily removed when the central line is removed, once the danger of Venous Thromboembolism has been resolved.
So to the big news that an unnamed hospital in the United Kingdom is currently using the catheter. Indeed hospital employees have reported already using the device to help save the life of one female patient in ICU.
Looking forward to more tales of success for the device, maybe a little more newsworthy than this one? Us too.
Source: San Antonio Business Journal