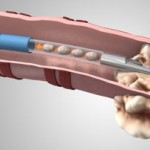
Particulate matter in the delivery system of Medtronic’s newly FDA approved CoreValve Evolut R system present enough risk to warrant and FDA Class 1 Recall
Medtronic
Medtronic’s New Biopsy System Targets Improved Lung Cancer Diagnosis
Medtronic tells us its recently FDA cleared system can get to difficult-to-access areas and collect larger, more intact samples
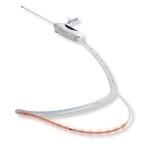
Data Supports Adhesive for Chronic Venous Insufficiency
12 month closure rates for the VenaSeal™ closure system comparable to radiofrequency ablation
Medtronic Launches GastriSail™ Bariatric Surgery Device
New Bariatric Surgery device makes the most common weight loss procedure in U.S. more consistent and efficient
FDA Approval and CE Mark for Medtronic’s Arctic Front Advance® ST Cryoballoon
Based on clinician feedback Medtronic’s third generation cryoballoon nudges ablative treatment of atrial fibrillation along
FDA Cleared for U.S. Launch: Medtronic’s Euphora™ Semicompliant Coronary Balloon
Following on the heels of its non compliant forebear, the Euphora semicompliant coronary balloon offers enhanced pushability
U.S. Launch of Medtronic’s HawkOne™ Directional Atherectomy System for PAD
Medtronic says its system, acquired with Covidien, treats all plaque morphologies including severe calcium, while streamlining procedural efficiency
FDA Approves CoreValve for Valve-In-Valve Use, Expanding Treatment Options
CoreValve becomes first TAVR device to be indicated for replacement of failed surgical heart valves, expanding the patient population that can benefit from this minimally invasive procedure
FDA Approves Medtronic Protégé™ GPS™ Stent for Iliac Artery Stenosis
Supporting study points to 96% nine month patency and 99% freedom from target vessel revascularization
Pivotal Studies of Resolute Onyx™ Drug Stent Starts in Patients with Small Coronary Vessels
Medtronic’s DES with CoreWire technology to be studied in wide spectrum of patients, starting with those with the smallest coronary arteries
Artifical Pancreas: Medtronic in Pole Position
New data is about to be presented that underlines Medtronic’s status as the leading developer of automated glucose monitoring and insulin delivery systems
Medtronic Enjoys First Fruit of Covidien’s Product Pipeline
Medtronic has received U.S. FDA approval for the Pipeline™ Flex embolization device, recently absorbed into its product catalogue with the acquisition of Covidien.
CE Mark for New Sizes of CoreValve® Evolut™R TAVI Offering
Now available in Europe in 26 mm and 29 mm sizes, Medtronic’s TAVI valves are delivered through a 14 French equivalent delivery system-the lowest profile on the market.
Deal Done: Medtronic and Covidien Are One
Medtronic plc says that it has successfully completed the previously announced acquisition of Covidien plc
FDA Approves Medtronic Drug-Coated Balloon for Peripheral Artery Disease in Upper Leg
Medtronic has gained U.S. FDA approval for its IN.PACT Admiral drug-coated balloon for the interventional treatment of peripheral artery disease (PAD) in the upper leg.
Medtronic Touts FDA Approval and Launch of Two New CRT Quadripolar Leads
Newly FDA approved, additional Attain Performa® Quadripolar Leads deliver more options so physicians can optimize delivery of Cardiac Resynchronization Therapy