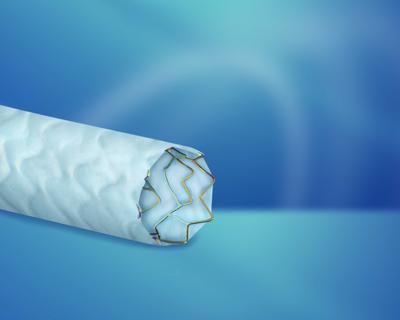
Biotronik’s PK Papyrus covered coronary stent system has received CE mark for the treatment of acute coronary artery perforation. The company is claiming 58% greater flexibility than Abbott’s Jostent Graftmaster due to its innovative “electrospun” membrane.
Background
Severe acute coronary vessel perforation during interventional procedures is a rare but catastrophic event that has shown up to 20% mortality according to research. One potential treatment is to exclude the defect by deploying a covered stent over the perforation. Many current covered stent designs require large ‘French size’ (F) guide catheters. Switching guide cathethers can cost the physician valuable time, delaying treatments. PK Papyrus is 5 F- and 6 F- compatible, thereby eliminating the need to switch access catheters during these emergency situations.
So how does it achieve this? It comes down to Biotronik’s clever “electrospinning” technology, which allows for a proprietary single-layer covered stent design, rather than the traditional “sandwich technique” used on comparable stents on the market. The single-layer covered stent design, with its 90µm polyurethane membrane, further enables a 24% lower crossing profile in comparison to Abbott’s Jostent Graftmaster, combining high flexibility and a low crossing profile for exceptional deliverability while sealing vessel defects in acute situations.
While PK Papyrus is based on the same proven Orsiro/PRO-Kinetic Energy stent platform, the wide size range (diameter 2.5-5.0 mm, length 15-26 mm) enables a broad range of vessels to be treated confidently and efficiently.
Physician comments
Prof. Georg Nickenig, University Hospital Bonn, commented: “In acute situations when there is a vessel perforation, I need a stent that is highly effective and has optimum deliverability. With PK Papyrus, I am confident that, if such a situation arises, I have a stent that is straight-forward to introduce into the artery with a flexibility that means simplified placement.”
Company comments
“PK Papyrus is a testament to the engineering excellence at Biotronik,” said Dr. Alexander Uhl, Vice President Marketing, Vascular Intervention, Biotronik. “The application of this electrospinning technology to the already highly deliverable Biotronik Orsiro/PRO-Kinetic Energy stent platform has allowed us to introduce a novel product to the market that will enable physicians to treat patients in emergency situations quickly and safely.”
Source: Biotronik SE & Co. KG
published: November 6, 2013 in: Approval/Clearance, Biotronik, Cardio