 ArtVentive Medical Group, Inc. has received European CE Mark certification for the ArtVentive EOS™ Peripheral Vascular Endoluminal Occlusion system.
ArtVentive Medical Group, Inc. has received European CE Mark certification for the ArtVentive EOS™ Peripheral Vascular Endoluminal Occlusion system.
Background
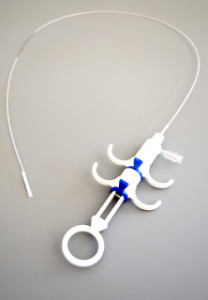
Californian company ArtVentive Medical Group, Inc. manufactures and markets a family of endoluminal occlusion devices (EOS).
The ArtVentive EOS™ device is catheter based and self-expandable serving for permanent or temporary occlusion of body lumens. Based on a proprietary technology platform the company is hoping EOS will provide an important new, minimally-invasive surgical option for the blockage of targeted veins, arteries and tubes within the body, with primary applications for peripheral vascular, neurology, cardiology, pulmonary and women’s health. In many cases they expect it will serve as a safe and reliable alternative to major surgery.
The EOS is catheter-based and employs flexible low-profile platforms as a delivery system. The device is offered in a range of diameters for different applications and can be placed in approximately five to ten minutes, depending on the surgery. It is anticipated this will reduce overall medical costs, and radiation exposure to patients and surgical teams and with a swifter recovery for the patient.
ArtVentive claims its device offers greater stability than current treatments. It reduces risk of vessel deflation via a means that also decreases the duration of the operation and causes a shorter segment of the vessel to be occluded. It is also applicable for closing tubular structures such as airways and reproductive tracts
The ArtVentive EOS device targets a substantive multi-billion dollar market demand in several major clinical areas including peripheral, neurovascular, women’s health (minimally invasive contraceptive device), interventional cardiology and pulmonary health issues.
Company comments
“This is a very significant achievement in the Company’s history as it moves toward commercialization and its planned launch into the European markets,” stated Dr. Leon Rudakov, President and CTO. “The CE Mark also paves the way to facilitate approvals for the ArtVentive EOS™ device internationally in meeting with the Company’s global mandate,” stated Jim Graham, CEO and Chairman.
Source: ArtVentive Medical Group, Inc., Business Wire
published: June 3, 2013 in: Approval/Clearance, Endoscopy
