ResMed has unveiled something called Air Solutions, which it is pitching as a sort of holistic approach to treatment of sleep disordered breathing.
Background
ResMed develops, manufactures and distributes medical equipment for treating, diagnosing, and managing sleep-disordered breathing, COPD, and other chronic diseases. It claims its innovative products and solutions are pitched at improving the health and quality of life of those who suffer from these conditions.
You immediately pick up the idea that ResMed is trying to invoke something of a paradigm shift in the treatment of sleep disordered breathing when you see it use the word “ecosystem” to describe its Air Solutions array of products. The company says its platform comprises components that, together, create this “comprehensive end-to-end ecosystem” that directly addresses the pain points for each stakeholder in the patient’s therapy, including physicians, sleep labs, home medical equipment providers, and the patients themselves.
The company would no doubt like us to explain the ecosystem concept which it says represents a new forward-thinking connected care solution for treating sleep-disordered breathing that can benefit all stakeholders in the care continuum. The ResMed Air Solutions platform has data-driven components that span from diagnosis to treatment, compliance management and patient engagement.The underpinning idea is that by tying all aspects of the care continuum together, efficiency and treatment efficacy will be enhanced by keeping providers informed and patients engaged.
Included in the Air Solutions platform are:
ApneaLink™ Air: ResMed’s compact and lightweight home sleep testing device works seamlessly with AirView software to empower sleep labs to efficiently manage each step of the diagnostic process, and share data to appropriate user groups.
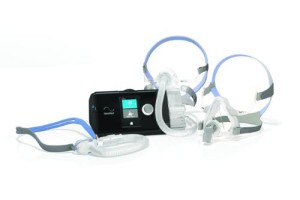
AirSense™ 10 Series: ResMed’s new series of continuous and automatic positive airway pressure devices (CPAP and APAP) called AirSense 10 that have advanced data and communications features to streamline the set-up, compliance management and compliance monitoring processes. This brand new device platform is also intuitive and easy-to-use, and offers a smaller bedside footprint.
AirCurve™ 10 Series: The AirCurve 10 series includes a range of bi-level devices that meet a variety of patient conditions and needs, such as patients who need extra pressure support, as well as an ASV device for patients with Cheyne-Stokes respiration or who require adaptive-servo-ventilation.
AirFit™ Masks: Announced earlier this year, the AirFit masks are known by their easy fit and superior comfort. The AirFit line includes the AirFit P10 nasal pillows system, which is the current market leader among new patients, the AirFit F10 full-face mask and the AirFit N10 nasal mask.
AirView™ Monitoring and Compliance Management System: When used in conjunction with Air Solutions devices, the AirView software solution significantly streamlines the patient management process by providing troubleshooting, fault detection and more. AirView enables data on demand, with patient data available in just one hour after a patient’s therapy session has ended.
myAir Patient Engagement Application and Software: An engaged patient is more likely to be compliant, and myAir helps patients better understand their own sleep data, with data dashboards, coaching and reinforcement messages.
ApneaLink Air and AirFit masks are available now in the U.S. and other select markets, says ResMed. The AirSense 10 and AirView will be available in the U.S. and Australia on Aug. 18, and other markets later this calendar year. The AirCurve 10 Series and myAir will also be available later this calendar year.
Company comments
“With Air Solutions, we’re creating a new beginning in patient care,” said Don Darkin, president of the ResMed Sleep-Disordered Breathing Business Unit. “As we unveil each Air Solutions component, our signature differentiation of superior performance and comfort will be underscored, made even stronger with groundbreaking technology that helps providers become more connected with their patients.”
Source: ResMed, Inc., PR Newswire
published: August 11, 2014 in: Healthcare, Launches / Withdrawals, Monitoring