In short
Californian spinal device company Benvenue Medical, Inc., has announced it has met the pre-determined stopping rules for enrollment completion in the KAST (Kiva System as a Vertebral Augmentation Treatment – A Safety and Effectiveness Trial) clinical trial.
Background
The National Osteoporosis Foundation estimates that there are 700,000 osteoporosis-related vertebral compression fractures annually in the U.S. alone, yet 200,000 kyphoplasty procedures are done globally.
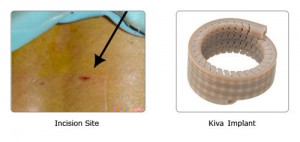
The Kiva VCF Treatment System provides an innovative approach to the treatment of painful Vertebral Compression Fractures (VCF). The Kiva VCF Treatment System features a proprietary flexible implant made from PEEK-OPTIMA®, a biocompatible polymer that is widely used and well accepted as a spinal implant. The Kiva Implant is designed to function as a mechanical support structure and a reservoir to contain and direct the flow of bone cement.
The Implant is delivered percutaneously in a continuous loop fashion into the vertebral body with an all-in-one disposable device through a small diameter, single incision. The amount of the Kiva Implant delivered can be physician-customised during the procedure to adjust to various fracture types. Delivered over a removable guidewire, Benvenue says its implant is designed to provide structural support to the vertebral body and to directionally control and contain bone cement.
The study
The KAST study is a randomised controlled trial comparing Kiva to balloon kyphoplasty, which is the current standard of care in the treatment of VCF. The trial enrolled patients at 21 medical centers in the United States, Canada, Belgium, France and Germany. Success will require non-inferiority on the primary endpoint, which is a composite of pain, function, and safety at one year of follow-up on patients treated on study. The study is designed to evaluate superiority on key secondary endpoints including PMMA cement volume, extravasation rate, and height restoration as well as other endpoints.
The KAST trial enrolled 300 patients and according to Benvenue is one of the largest randomised VCF studies to date studying the device versus balloon kyphoplasty, the current standard of care.
Investigator comments
“The KAST study enrolled ahead of schedule, and we believe the positive response we received is due to our investigators’ enthusiasm for a new treatment option for painful and debilitating osteoporotic vertebral fractures moving away from traditional vertebroplasty or balloon-based vertebral augmentation,” said Sean M. Tutton, MD, FSIR, Co-Principal Investigator in the KAST Study and Professor of Radiology and Surgery at the Medical College of Wisconsin in Milwaukee.
“We appreciate the efforts of the investigators and research coordinators, as well as the spine community’s enthusiasm to evaluate the potential benefits of the Kiva System. The results will be important to guiding treatment recommendations for VCFs,” said Steven R. Garfin, MD, Co-Principal Investigator of the KAST Study and Professor and Chairman of the Department of Orthopaedic Surgery at the University of California, San Diego Medical Center.
Company comments
“We continue to work very closely with the FDA, and we are pleased with the collaboration and progress we have made working together,” said Robert K. Weigle, CEO of Benvenue Medical, Inc.
Regulatory status
The Kiva device is CE marked and available in Europe through Zimmer Spine. The KASt trial is intended to support an FDA 510(k) submission when complete.
Source: Benvenue Medical, Inc., PR Newswire
published: July 31, 2012 in: Clinical Studies/Trials, Spine, Techniques
