 Endovascular repair (EVAR) device company Aptus Endosystems, Inc., has announced that its ANCHOR post-market registry enrollment has surpassed its key milestone of 250 patients.
Endovascular repair (EVAR) device company Aptus Endosystems, Inc., has announced that its ANCHOR post-market registry enrollment has surpassed its key milestone of 250 patients.
Background
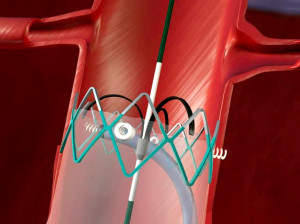
We’ve covered Aptus’ HeliFx™ quite extensively over the past few years as the system has leapt over numerous regulatory hurdles around the world. In essence the system of helical anchors can be used during primary EVAR procedures to enhance an endograft’s inherent fixation and sealing mechanisms. Doing so can potentially improve the long-term durability of the aneurysm repair. The system can also repair endovascular grafts that have developed endoleaks, migrated away from the implant site, or are at risk of developing these complications, which are often seen after EVAR. In such cases, augmented fixation and/or sealing is required to regain or maintain effective aneurysm exclusion.
Tagged ANCHOR (Aneurysm Treatment Using the Heli-FX Aortic Securement System Global Registry), the data collection has come from a global, multicenter, prospective post-market registry set up to evaluate the use of the Heli-FX™ System in EVAR and capture critical data on use of the technology and acute and long term procedural outcomes.
Physician comments
US Principal investigator William Jordan, M.D., Chief, Section of Vascular Surgery at the University of Alabama Hospital, Birmingham, Ala., stated; “As the use of this new tool to secure endografts becomes more widespread, we are gaining scientific validity of these results in the real world setting. Clinicians from around the world are contributing their experiences to help us understand the best application of the Heli-FX System,”
European lead, Jean Paul de Vries, M.D., Ph.D., Head of the Department of Vascular Surgery at St. Antonius Hospital, Nieuwegein, the Netherlands said; “The ANCHOR Registry will provide real world data to substantiate the benefits of the Heli-FX System. This data will give us answers regarding the prevention of neck dilatation post-EVAR.”
Kenneth Ouriel, M.D., former Chief of Surgery at Cleveland Clinic said; “The ANCHOR findings suggest that the Aptus device will play a prominent role in the treatment of AAA, expanding the number of patients who may be treated with a minimally-invasive solution and providing a suitable option for those with a previously placed endograft that has developed a leak or migration,”
Company comments
“The rapid acceptance of the Heli-FX technology by the ANCHOR investigators allowed us to reach this 250 patient milestone well ahead of our plans,” said James Reinstein, President and CEO of Aptus Endosystems, Inc. “We are impressed by the early technical success of the technology. Our expectation is to see a reduction in secondary interventions when EndoAnchors are used in these difficult to treat anatomies.”
Source: Aptus Endosystems, Inc., Business Wire
published: July 11, 2013 in: Clinical Studies/Trials, Vascular
