In short
Back in January we reported FDA’s 510(k) clearance of TriReme Medical, Inc.’s Chocolate PTA balloon catheter, here. Now the company has announced that it has received FDA clearance for an expanded matrix of sizes of its tantalisingly named device. Chocolate™ is now approved for the percutaneous transluminal treatment of lesions in the peripheral vasculature in balloon diameters from 2.0 – 4.0mm and in balloon lengths up to 120mm.
Background
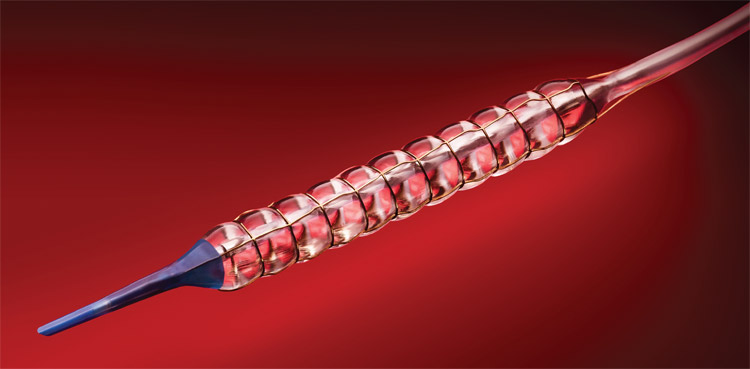
Chocolate’s novel design incorporates a nitinol constraining structure over a semi-compliant balloon to facilitate the formation of small modules (“pillows”). Through this mechanism of action, Chocolate minimises shear stress and allows for uniform inflation and rapid deflation. The Chocolate “pillows” can expand locally to facilitate plaque modification and are designed to lower the strain and trauma induced on the vessel wall by ensuring the balloon cannot inflate beyond the extent of its constraining frame at any point.
Company comments
“This latest regulatory approval broadening the available size matrix of the Chocolate™ PTA Balloon marks yet another important milestone for TriReme Medical,” said Eitan Konstantino, President and CEO of TriReme Medical Inc. “There is more to come; our goal is to provide a broad range of innovative tools to improve outcomes in patients suffering from complex vascular disease.”
Clinician comments
“Chocolate™ has become an essential part of my routine practice for treating below the knee lesions,” said Dr. Rajesh Dave, Chief Medical Executive and Director at Cardiac Catheterisation Laboratories, Holy Spirit Cardiovascular Institute, Camp Hill, PA. “With the approval of these new sizes, I will be able to expand my use of Chocolate™ and begin to treat patients with disease in the popliteal artery where we usually prefer not to stent.”
Source: Trireme Medical Inc., PR Newswire
published: June 20, 2012 in: Approval/Clearance, Vascular
