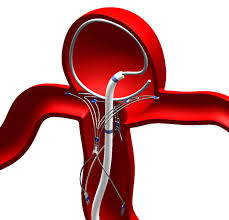
Pulsar Vascular has announced U.S. FDA approval of an Investigational Device Exemption (IDE) for its PulseRider®, a minimally invasive, aneurysm neck reconstruction device. Trial will follow.
Background
The IDE allows Pulsar Vascular to begin a multicenter clinical trial in support of a Humanitarian Device Exemption (HDE) to evaluate the PulseRider for U.S. approval for wide neck aneurysms at or near a bifurcation of the basilar tip or carotid terminus. There are currently no devices approved by the FDA for this indication and a flick through the pages of Pulsar Vascular’s illuminating website will spell out just how clever this thing is. In essence it is a repositionable, retrievable nitinol scaffold that holds the aneurysm neck open in order for blood to flow to the desired vessel during introduction of an embolic device such as a coil into the aneurysm itself.
The PulseRider received CE Mark in late 2013 and it has been in use in Europe since early 2014. The company says its device addresses an unmet clinical need to treat complex necked bifurcation aneurysms, and the European physicians have welcomed it into their practice. It plans to expand the European market while the US clinical trial is underway.
Physician comments
Dr. Adnan Siddiqui, at the University of Buffalo said, “The treatment of bifurcation aneurysms is truly an unmet need in endovascular therapies. I am excited by the positive response to the PulseRider in Europe, and as part of the US clinical trial, I look forward to working with the company and the FDA, to bring this novel, innovative technology into my everyday practice.”
Company comments
Rob Abrams, the CEO of Pulsar Vascular and co-creator of the PulseRider design, said, “The FDA’s approval of the PulseRider IDE allows us to initiate this important study for our flagship product and validates our scientific platform technology. This approval represents another significant milestone for Pulsar Vascular and a step forward on the path to providing a new treatment option to both patients and physicians.” The clinical trial will be conducted at multiple clinical sites in the US and is slated to begin Q3 2014.
Source: Pulsar Vascular, PR Newswire
published: June 10, 2014 in: Approval/Clearance, Clinical Studies/Trials, Neuro, Vascular