In short
Back in early October, Californian cardiovascular company TriVascular announced that it had been granted approval under an FDA to market its Ovation Stent Graft. Now the FDA has issued its own confirmatory press release on the subject.
Background
We covered the approval of Ovation as a Humanitarian Use Device (HUD) around this time last year. Since then we have also followed the device’s progress, including FDA’s issuance of guidance for users back in March of this year, here.
Humanitarian Use Device approval is limited to those devices that treat or diagnose fewer than 4,000 people in the United States each year. This newly issued PMA now means the device can be marketed according to its indications on a fully commercial basis.
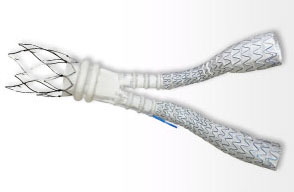
Treatment with a stent graft depends on the patient’s anatomy. A small number of people have blood vessels that are too small in diameter to accommodate typical endograft systems. The 20mm diameter Ovation Abdominal Stent Graft System, is not only small in diameter but it uses a narrower delivery system than any other marketed endograft (4.7 mm in diameter compared to 7 mm in diameter). In this way the Ovation system provides patients with small arteries the option of less invasive surgery to repair their potentially life-threatening abdominal aortic aneurysm.
The way it achieves this feat is through the replacement of a portion of the traditional metal stent with ring-shaped channels. After the device is in place in the aorta, the channels are injected with a polymer, expanding the endograft against the aorta to create a seal.
“I am proud to have the opportunity to work with the internationally recognized vascular
experts that participated in this trial and treated a global cohort of patients with abdominal
aortic aneurysms,” said Manish Mehta, MD, MPH, Director of Endovascular Services at The
Vascular Institute for Health & Disease in Albany, New York, who served as the Principal
Investigator for the company’s pre-approval study.
“The 1-year clinical trial outcomes, particularly in patients with complex anatomies, demonstrate the excellent safety and effectiveness of the Ovation stent graft system. With Ovation’s FDA approval, we can confidently offer EVAR to more patients since we now have access to a very safe and effective low profile system.”
“The unique construction of the 20-mm Ovation system allows it to fit into a narrower delivery catheter than other endografts currently on the market. FDA’s approval of this product will enable some patients with small blood vessels to be treated via minimally-invasive surgery who did not previously have this option,” said Christy Foreman, director of the Office of Device Evaluation in the FDA’s Center for Devices and Radiological Health.
Source: U.S. Food and Drug Administration
published: November 13, 2012 in: Approval/Clearance, Vascular
