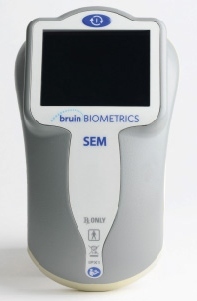
Bruin Biometrics, LLC has gained CE Mark approval to market the SEM Scanner in the European Union. The SEM Scanner is that is intended to detect localized tissue edema and pressure induced tissue damage that may lead to the formation of pressure ulcers (also known as pressure sores, or decubitus ulcers) before damage becomes visible on the skin’s surface.
Background
The hand-held, portable SEM Scanner is a a Class IIA medical device intended for point of care diagnosis of tissue damage. It measures increased fluid content within the skin and underlying tissue known as subepidermal moisture (SEM), which can serve as a biophysical marker of inflammation indicative of impending skin damage and pressure ulcer formation. In clinical studies, SEM has been shown to identify pressure-induced tissue damage — including pressure ulcers — up to 10 days before damage becomes visible on the skin’s surface.
Early detection of pressure ulcers with the SEM Scanner can lead to targeted interventions, reduced ulceration severity, and early recovery, as well as decreased hospital days. Additional advantages of the SEM Scanner over other diagnostic modalities include objective, evidence-based measurements; non-invasive, rapid results; clear risk assessment documentation; standardized patient assessment; and minimal technical skills for operation.
BBI developed the SEM Scanner in conjunction with research colleagues at the University of California at Los Angeles (UCLA), notably Barbara Bates-Jensen, PhD, RN, CWOCN, FAAN. Bates-Jensen serves as Chair of the Faculty Executive Committee at the UCLA School of Nursing and Associate Adjunct Professor, Medicine at the UCLA David Geffen School of Medicine.
BBI will officially launch the SEM Scanner at the upcoming MEDICA conference taking place in Dusseldorf, Germany, November 20-23, 2013.
Source: Bruin Biometrics, LLC., PR Newswire
published: November 14, 2013 in: Approval/Clearance, Launches / Withdrawals, Wound Care