In short
Medtronic, Inc. has announced that the Sentrant Introducer Sheath has received the CE mark and will soon be launched internationally.
The new device complements Medtronic’s portfolio of stent grafts for the endovascular repair of aortic aneurysms and related conditions.
Background
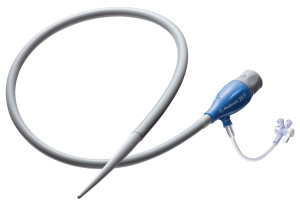
The Sentrant Introducer Sheath is designed for use with Medtronic’s Endurant II AAA and Valiant Captivia Stent Graft Systems, the market-leading devices in their product categories worldwide, and can also be used with similar devices for endovascular repair. Inserted at the access site in the patient’s femoral artery, the tapered tube is advanced up into the iliac arteries to facilitate the implant procedure, enabling smooth passage of the stent graft delivery system and accessory devices en route to the treatment site in the aorta.
Medtronic claims its Sentrant Introducer Sheath offers an optimal seal compared to competitive sheaths for superior hemostasis during the procedure. Furthermore it’s reinforced coil is kink resistant and its hydrophilic coating and flexibility allow easy tracking through tortuous and calcified iliac vessels.
To accommodate a range of anatomies, the Sentrant Introducer Sheath is made in diameters of 12–26 French and lengths of 28cm and 64cm.
Compatible with the Archer Super Stiff Guidewire and the Reliant Stent Graft Balloon Catheter, it expands Medtronic’s accessories toolkit for endovascular aortic interventions.
Medtronic’s complete portfolio of stent grafts and related accessories including the new introducer sheath will be on display during the Charing Cross symposium in London this weekend.
Source: Medtronic, Inc.
published: April 5, 2013 in: Approval/Clearance, Congresses and Meetings, Medtronic, Vascular