In short
In a press release issued by PR Web, Ohio based Endosphere Inc. has reported that its new, incisionless, implantable antiobesity medical device called Satisphere has gained CE Mark clearance.
Background
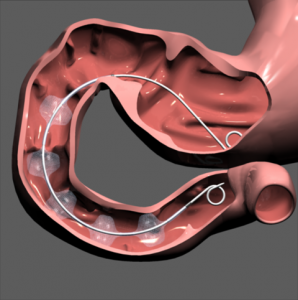
Rather like GI Dynamics’ Endobarrier®, this is another device aimed at treating obesity and type 2 diabetes, aiming at the same result of reducing calorific intake in the duodenum, albeit by adopting a different strategy.
The Satisphere device works by actually slowing the passage of food through the duodenum, the upper part of the smaller intestine that breaks down food. Because food is in contact with the duodenum for a longer time, the device essentially fools the body into thinking that it’s consumed more food than it actually has.
Like the aforementioned Endobarrier, the device can be inserted endoscopically and does not require surgery or medication.
The device can be placed in the small intestine in about 15 minutes, taken out in five minutes, if necessary. It doesn’t require any suturing or hooks to hold it in place.
Endosphere claims the ability to quickly and easily remove the device if a patient no longer needs it is one of its chief advantages.
Availability
Now that the device has been approved for European sales, the company’s next big step is to start selling it. EndoSphere is already in discussions with potential customers, said Chris Thorne of Broadline Capital, who serves as EndoSphere’s executive chairman.
There’s no time line on an application for U.S. Food and Drug Administration approval of the device, Thorne said.
Source: Endosphere Inc., PR Web
published: May 1, 2012 in: Approval/Clearance, Endoscopy, Gastroenterology, News, Techniques
