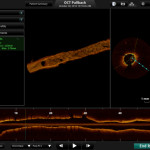
Elixir Medical has seen one year results from its pivotal study presented at TCT2013. And impressive they are too, with luminal area sustained and low MACE rate.
Congresses and Meetings
Abbott Gets Timing Right with Mitraclip® Approval and New Clinical Support at TCT2013
FDA’s Mitraclip approval now means it’s available in the United States for patients with significant symptomatic degenerative MR who are at prohibitive risk for mitral valve surgery. New data at TCT2013 provides compelling clinical support for the device in this patient group.
Surgical Innovations Hosts UK’s First International ‘Ultra’ MIS Congress
Surgical Innovations (SI), the UK-based designer and manufacturer of Minimally Invasive Surgery(MIS) devices, hosted its first ever international ‘Ultra’ MIS congress on Friday October 25th. Surgeons attended the event from across the UK and Europe.
Lower Mortality Rate in STEMI Patients with MGuard Stent at Twelve Months
At TCT2013 this week InspireMD, Inc., has announced new 12-month study data which, it says, demonstrates that the MGuard outperformed bare metal and drug eluting stents in all-cause mortality in ST segment elevation myocardial infarction (STEMI) patients.
18-month Average 24 mmHg BP Reduction Reported with St.Jude EnligHTN™ Renal Denervation System
Data presented at the TCT2013 symposium indicates that patients with drug-resistant hypertension treated with the EnligHTN™ Multi-Electrode Renal Denervation System averaged a 24 mmHg reduction in systolic blood pressure as measured in an office setting.
Direct Flow Medical® TAVI System Sees Strong Six Month Data Presented at TCT2013
Transcatheter heart valve innovator, Direct Flow Medical®, Inc., has seen six month implant data presented at the TCT2013 congress, from the now fully-enrolled DISCOVER CE Mark Trial. A company press release says the results continue to affirm the excellent outcomes and strong safety profile of the company’s Transcatheter Aortic Valve System.
ART’s “Dismantling” Stent in Good Shape at 30 Days, Says Data Presented at TCT2013.
Arterial Remodeling Technologies has seen early data concerning its “dismantlable” temporary coronary stent presented at TCT2013. It seems the device is maintaining its integrity at 30 days, with low vessel recoil rate akin to metallic stent performance. Next data points will be more interesting, however reassuring this early data is.
MiStent SES 2 Year Findings Show Sustained Prevention of Lumen Loss
Micell’s MiStent SES is a unique concept in which the eluting drug outstays the carrier polymer. The theory is that this should afford longer term protection against inflammation and stenosis, and new data, presented at TCT2013, provides clinical support for the claim.
Infraredx Breakthrough Coronary Artery Imaging Showcased at TCT 2013
Infraredx, Inc. describes its TVC Imaging System™ as being capable of the true vessel characterization of coronary artery disease. The system will be highlighted in 13 presentations during the annual Transcatheter Cardiovascular Therapeutics Meeting which starts this Sunday in San Francisco.
US Launch of St.Jude’s ILUMIEN™ OPTIS™ 3-D Vessel Reconstruction Technology
TCT2013 will see St. Jude Medical, Inc. showcasing its newly FDA approved ILUMIEN™ OPTIS™ PCI Optimization™ System, a new technology designed to provide physicians with a comprehensive disease assessment tool for treating patients with coronary artery disease.
Study Results Support AtheroMed’s Phoenix Peripheral Artery Disease Atherectomy System
Vascular device specialist AtheroMed, Inc., says its Endovascular Atherectomy Safety and Effectiveness (EASE) study demonstrates the safety and effectiveness of the Phoenix Atherectomy System in treating PAD. Results were presented at the Vascular Interventional Advances (VIVA) 2013 conference in Las Vegas in early October 2013.
Covidien’s New Clinical Data is a Statement of Intent for its Peripheral Arterial Disease Business
Covidien has released positive final results from its DURABILITY II study and promising preliminary data from its DEFINITIVE AR Peripheral Artery Disease (PAD) trial involving some of its technologies. The results are sure to fuel its ongoing push into the peripheral vascular therapy area.
New Studies Say SmartPill® Delivers Window into Gastrointestinal Motility Disorders
Two new studies on Given imaging’s SmartPill® device provide an insight into the usefulness of this technology in understanding gastrointestinal motility. The papers have been presented as posters at the American College of Gastroenterology’s 2013 Annual Scientific Meeting (ACG).
Newly Cleared Integra® Hollywood™ NanoMetalene™ Interbody Device Brings Best of Both Worlds
Ingeniously, the newly FDA cleared Hollywood Nanometalene IBD from Integra comprises a PEEK-OPTIMA core construct, coated with an ultrathin layer of titanium, providing the best of both worlds in Interbody Device design.
NASS Launch for Invuity® BREITEN™ Illuminated Retractor Offers “Unsurpassed” Anterior Cervical Disc Space Visualization
At this week’s North American Spine Society (NASS) Annual Meeting the company has introduced its Breiten™ Illuminated Retractor System. The system promises to increase surgical efficiency, reduce operating room time and lessen surgeon fatigue by eliminating or reducing dependence on a headlight
AAOMS 2013 Sees Depuy Synthes CMF Launch Integrated, Customized Facial Reconstruction
TRUMATCH® CMF Solutions Spans Preoperative
Virtual Surgical Planning Services, Intraoperative Tools and Customized Implants