Italian cardiovascular device maker Sorin Group, has gained U.S. FDA approval for the Solo Smart™ Aortic Pericardial Heart Valve which the company claims is the first biological valve to feature a fully removable stent.
Sorin
Sorin’s Phospholipid-Reducing Crown Valve Gains CE Mark
Cardiovascular device specialist Sorin Group, has gained CE mark certification for its innovative CROWN PRT™ stented aortic bioprosthesis.
FDA Approval for Sorin’s Phospholipid-Reduced Mitroflow™ Aortic Heart Valve
Italian Cardiovascular company Sorin Group, has received U.S. FDA approval for the Mitroflow Aortic Pericardial Heart Valve with Phospholipid Reduction Treatment (PRT).
Sorin’s Perceval™ Sutureless Aortic Valve Now CE Marked for Under 65s
Italian Cardiovascular device specialist Sorin Group tells us its Perceval™ sutureless aortic valve has received CE mark approval for adult age indication allowing treatment of a wider spectrum of patients with aortic stenosis and/or steno-insufficiency.
Sorin Buys Oscor’s CRM Lead Business Including Manufacturing Facility
Italian Cardiovascular device company Sorin Group has announced the purchase of the Oscor Inc. lead business, including a lead manufacturing facility in the Dominican Republic for an aggregate value of approximately $20 million (Eur15.4 million).
Sorin Doesn’t Let an Earthquake Halt Profit Progress
Sorin’s financials for 2013 look pretty punchy and despite a few product-lifecycle challenges appear to indicate that the financial impact of the recent earthquake experienced at one of the company’s facilities are well behind it.
Sorin Group Launches Intensia Family of ICD and CRT-D Devices Featuring DF-4 High Voltage Connector
Italian Cardiovascular device maker Sorin Group has gained CE Mark approval for its INTENSIA family of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy (CRT-D) devices, all of which feature a DF-4 high voltage connector.
CE Mark and First Implant of Sorin’s KORA 100 MRI-Compatible Pacing System
Italian Cardiovascular device company Sorin has confirmed that it has gained CE Mark approval and seen a first implant of the KORA 100 pacing system, which is MRI compatible.
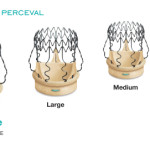
CE Mark for Extra Large Sizes of Sorin’s Perceval™ Sutureless Aortic Valve
Last week we covered the news that Italian cardiovascular outfit Sorin had gained CE mark approval for its Solo Smart stentless heart valve. Now the company tells us it has also gained the European nod for the large sizes of its sutureless Perceval valve.
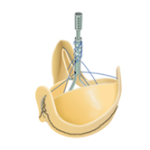
CE Mark and First Implant for Sorin’s “Removable Stent” Biological Valve
Sorin gets CE mark approval for Solo Smart,the evolution of Freedom Solo, a stentless and biological aortic valve with no synthetic material, proven outstanding hemodynamic and clinical performance.
Sorin’s Q3 Revenues Show Recovery from 2012 Earthquake
Q3 revenues for Sorin are somewhat clouded by last year’s earthquake which saw supply of some of its core lines temporarily suspended. A year later and the Italian cardiac device maker is recovering nicely as revenues return to pre-disaster levels and above.
Sorin and Boston Scientific in TAVI Manufacturing Tie-up
Italy’s Sorin Group has issued a press release in which it confirms it will be participating in the manufacture of … continue reading “Sorin and Boston Scientific in TAVI Manufacturing Tie-up”
FDA Approval For Smartview™ Means Sorin ICD Patients Can Be Monitored Remotely
Sorin’s next-generation implantable defibrillators can now be monitored remotely using the company’s Smartview technology which has now gained FDA approval.
Conditional FDA Approval For Sorin’s RESPOND CRT Trial
Sorin’s newly approved RESPOND CRT trial aims to demonstrate clinical benefit of innovative SonR® CRT optimization system in cardiac resynchronization therapy.
Sorin Bids For A Seat At Percutaneous Mitral Valve Table
Sorin’s investment in HighLife SAS will fund development of an innovative percutaneous mitral valve replacement technology, over which the Italian heart company will retain an option to buy.
Sorin Acquires Belgian Nerve Stimulator Company, NeuroTECH
A spin-out from a Belgian University has been snapped up by Sorin, in a deal which looks like it’s intended to give the Italian Cardiovascular company a toe-hold in vagus stimulation therapies for heart failure patients.