FDA approval for SynCardia temporary Total Artificial Heart with SynHall valves, gives the company control over the last key component to manufacture the Total Artificial Heart.
Products
EXCOR® Pediatric Ventricular Assist Device Study Completes Enrollment
Berlin Heart’s Humanitarian Device Exemption for its Excor Pediatric VAD was conditional on undertaking a post-approval study. Now enrollment is complete and most patients have reached endpoint date.
Ethicon Launches HARMONIC FOCUS®+ Shears with Adaptive Tissue Technology
Ethicon Endo-Surgery says its newly launched device offers surgeons improved thermal management coupled with greater precision and speed when performing resection during open procedures.
Novel Advanced Parkinson’s Pharma Therapy Delivery Jumps EU Eligibility Hurdle
Israeli pharma company NeuroDerm is making progress towards approval of a subcutaneously delivered therapeutic candidate for advanced Parkinson’s Disease.
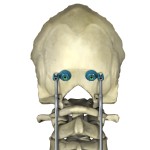
FDA Nod for Medicrea K-JAWS® Cervical Fixation Staple
Medicrea says its K-Jaws spinal cervical compression staple offers a less invasive and quicker solution than cervical fixation option.
Complex Spine and MIS Line Extensions & Regulatory Clearances for K2M
K2M has gained U.S. marketing clearance for its CASPIAN® Occipital Anchor Spinal System and has also announced other product introductions across the globe.
Codman Neuro’s New Envoy® Guiding Catheters, Target Access, Support and Stability
Codman Neuro has chosen the SNIS meeting at which to launch the ENVOY® DA XB Distal Access Guiding Catheter and the 7F ENVOY® Guiding Catheter for neurovascular procedures.
CE Mark for Boston Scientific’s Agent™ Drug-Coated Coronary Balloon
Boston Scientific’s Agent™ Paclitaxel-Coated PTCA Balloon Catheter has gained CE mark approval and will now be launched in Europe.
FDA Approves Sorin’s Solo Smart™, First Stentless Fully Biological Valve
Italian cardiovascular device maker Sorin Group, has gained U.S. FDA approval for the Solo Smart™ Aortic Pericardial Heart Valve which the company claims is the first biological valve to feature a fully removable stent.
FDA Clears 150 cm Arrow® GPSCath® Balloon Dilatation Catheters
Teleflex subsidiary Hotspur Technologies has gained FDA clearance for its 150cm length Arrow GPSCath dual function balloon dilatation catheters.
Newly Approved Prestige® LP Moves Cervical Disc Game On
Medtronic has based its new device on the design principles of its successful predecessor, but a few changes of approach and material represent evolutionary steps.
FDA Clears PicoSure™ for Treatment of Acne Scars
Cynosure, Inc., has received FDA 510(k) clearance for its flagship PicoSure Picosecond Laser Workstation for the treatment of acne scars.
Sorin’s Phospholipid-Reducing Crown Valve Gains CE Mark
Cardiovascular device specialist Sorin Group, has gained CE mark certification for its innovative CROWN PRT™ stented aortic bioprosthesis.
CE Mark and First Use of St. Jude’s FlexAbility™ Ablation Catheter
St. Jude Medical, Inc. has gained CE Mark approval and announced first use of the FlexAbility™ Ablation Catheter, a novel ablation technology used by electrophysiologists for the treatment of cardiac arrhythmias.
FDA Clears FLXfit™, The World’s First 3D Expandable Interbody Cage
Expanding Orthopedics Inc., has announced that its FLXfit™, the world’s first 3D expandable interbody cage, has gained U.S. FDA 510(k) clearance for commercialization in the US.
FDA Approves Boston Scientific’s REBEL™ Coronary Stent
U.S. FDA approval of REBEL™ Platinum Chromium Coronary Stent brings the benefits of modern stent architecture to patients not suitable for drug eluting variants.