System offers potential to reduce radiation exposure during orthopedic imaging in patients requiring repeat visits
Regulatory
FDA Nod Means Shared Glucose Monitoring Reality Using Apps
Now people with diabetes as young as 2 years of age can automatically and securely share real-time CGM data from their Dexcom receiver to a caregiver’s mobile app
First MRI-Conditional Cochlear Implant Approved in U.S.
MED-EL USA has announced U.S. FDA approval of its new SYNCHRONY cochlear implant, the first time such a device has been approved for use with 3.0 Tesla MRI without the surgical removal of the internal magnet.
CE Mark and Launch for AliveCor AF Detection Function
AliveCor, Inc. has received CE Mark clearance for an automated atrial fibrillation detection function on its AliveCor® Heart Monitor.
FDA Clears K2M’s New Mesa® 2 Pedicle Screw System
Mesa 2 claims quicker on/off capability and one-step locking.
FDA Clearance for Handheld Bladder Scanner
Medical device company, Signostics, has gained U.S. FDA (510(k) clearance for its SignosRT Bladder, the company’s new hand-held bladder scanner.
Good News for BPH Sufferers as MIS Embolization Procedure Gains CE Mark
“Prostatic Artery Embolization offers a safe and effective treatment option for men.”
CE Mark for Silk-Enhanced Biological Bone Graft
Silk from moths enhances cohesion and mechanical properties of new bone graft
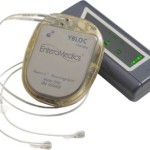
FDA Approves First Obesity Treatment Device for Ten Years
Enteromedics is celebrating approval of its novel neuroscience-based technology that it says enables safe, durable eeight loss
Next Patient Please: InVivo Scaffold Study Restarts Recruitment
InVivo Therapeutics has been given the green light to reopen subject enrollment for the pilot trial of its investigational Neuro-Spinal Scaffold in patients with acute spinal cord injury
FDA Clears Valorem Surgical® MaxiMIS™ Spinal Fixation System
Valorem Surgical has received FDA clearance and seen a first U.S. case utilizing its minimally invasive MaxiMIS™ Spinal Fixation System.
FDA Clears t:flex™ Largest Capacity Insulin Pump
Insulin pump maker, Tandem Diabetes Care®, Inc. has gained U.S. FDA clearance to market the t:flex™ Insulin Pump, with its 480 unit reservoir.
Expanded Clearance for Intact® Tissue Excision System
Intact Medical Corporation’s marketing clearance sees it usable for remove intact breast lesions up to 30mm in diameter.
FDA Clears PorOsteon’s Phusion Metal™ Nitinol-Based Cervical Cage
The U.S. FDA has cleared new nitinol-based Phusion Metal™ Cervical Cage which aims to maximize bone growth through a fully interconnected porous structure, while matching the modulus of living cancellous bone.
FDA Inks De Novo Approval for VasoPrep Surgical’s Surgical Marker Pen
VasoPrep Surgical tells us that the U.S. FDA has cleared its surgical marking pen under the agency’s de novo pathway
FDA Approves Medtronic Drug-Coated Balloon for Peripheral Artery Disease in Upper Leg
Medtronic has gained U.S. FDA approval for its IN.PACT Admiral drug-coated balloon for the interventional treatment of peripheral artery disease (PAD) in the upper leg.