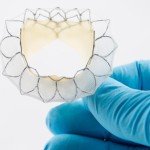
With its newly announced CE mark, Tryton Medical has become the purveyor of the first & only coronary bifurcation stent indicated for Left Main Coronary artery application.
Cardio
U.S. Launch of Endologix VELA™ Proximal Endograft System
The U.S. launch of a new endograft system from Endologix has taken place with the company claiming its novel VELA stent graft provides enhanced visibility, control, and precision in placement.
Sorin Doesn’t Let an Earthquake Halt Profit Progress
Sorin’s financials for 2013 look pretty punchy and despite a few product-lifecycle challenges appear to indicate that the financial impact of the recent earthquake experienced at one of the company’s facilities are well behind it.
FDA’s Over-the-Counter Clearance for AliveCor Heart Monitor
The AliveCor™ Heart Monitor is a single-channel ECG recorder that works with your iPhone or android device to record,store and transfer heart data for people requiring monitoring. Previously cleared for issue by prescription only, the device is now available for Over the Counter pre-order purchase, with shipments beginning in March.
First US Patient Receives Nanostim™ Non-Surgical, Leadless Cardiac Pacemaker
St. Jude Medical, Inc. has announced the first U.S. implant in the company’s LEADLESS II pivotal trial designed to evaluate the Nanostim™ leadless pacemaker on its way to U.S. FDA approval.
Edwards’ SAPIEN XT Transcatheter Valve CE Marked for Mitral and Aortic Valve-in-Valve Procedures
Edwards Lifesciences Corporation has received CE Mark approval for the use of its SAPIEN XT transcatheter heart valve for valve-in-valve procedures, where failing bioprosthetic valves require intervention.
Eventful 2014 Expected for Thoratec® Heart Assist Devices
While Thoratec’s numbers landed within their guidance figures, we somehow expected to see more from a company that purports to be changing the face of the heart-assist device world. The mood music suggests they’re readying themselves for acceleration beyond 2014 though.
Successful First Human Implant of Neovasc Tiara™ Transcatheter Mitral Valve
Minimally invasive mitral valve surgery has taken a step forward with the report of a first in human use of Neovasc’s Tiara transapical implant. Everything went well and no residual paravalvular leakage is reported.
Aneurysms Holding at 2 Years in Cordis INCRAFT® AAA Stent Graft Study
J&J Subsidiary, Cordis Corporation has seen two-year data from its INNOVATION Trial presented at the 2014 Leipzig Interventional Course in Germany. Data from the study continues to demonstrate that the INCRAFT® System performs well in patients suffering from abdominal aortic aneurysms (AAA) two years after treatment.
Impella Propels Abiomed to 21% Revenue Growth Over Equivalent Quarter Last Year
Buoyed by healthy revenue growth from its Impella in-dwelling heart-assist device, Abiomed has reported a Q3 top line up 21% compared with the same period a year ago. Profits are also up with Q3 GAAP net income of $4.4 million compared to $2.7 million last year.
Edwards Lifesciences Sapien 3 Heart Valve to Launch Imminently Following E.U. Approval
Edwards Lifesciences Corporation has received CE Mark approval for the SAPIEN 3 valve. European launch of what Edwards is calling its most advanced transcatheter aortic valve is imminent.
First UK Patient for Tiny Leadless Nanostim™ Pacemaker
St. Jude Medical, Inc. is trumpeting the first post-approval implant of the Nanostim™ leadless pacemaker in the U.K. The world’s first retrievable leadless pacemaker was implanted at St. Bartholomew’s Hospital in London, UK, by Professor Richard Schilling.
FDA OK’s Medtronic Pacemakers for Full-Body MRI Scans
Medtronic, Inc. has announced the U.S. FDA approval of its Medtronic SureScan® pacing systems that are compatible with use of MRI scanning on any region of the body.
Study Points to Savings with Remote Monitoring of ICD Patients
Biotronik is pointing to new study data showing that use of its Biotronik Home Monitoring® system has saved the French National Health Insurance system costs, in addition to being safe and efficient.
FDA Clearance Means OptionELITE IVC Filter Becomes First Over-The-Wire Retrievable System
Argon Medical Devices, Inc. says it has received FDA clearance to begin marketing its OptionELITE retrievable inferior vena cava filter with a new over-the-wire delivery technique. This new clearance enables physicians to safely deliver the filter to a patient’s IVC by following the path of a guidewire.
On-X® Mechanical Heart Valve Becomes First to Officially Need Lower Blood-Thinner Dose
New clinical data has been used to support a modification in the CE Mark labelling claims of the On-X® Life Technologies Inc. mechanical aortic valve, which means patients will require significantly lower doses of blood thinner than other mechanical heart valves.