St. Jude Medical, Inc. has gained CE mark approval for a 25 mm iteration of its Portico™ Transcatheter Aortic Heart Valve Implantation System, extending the applicability of the device to more patients for whom transcatheter aortic valve replacement (TAVR) is indicated.
Cardio
Panel’s Yes Vote Means FDA Will Find it Hard to Say No to Boston’s WATCHMAN™ LAA Closure Device
Could the FDA be about to approve a permanently indwelling, percutaneously inserted Left Atrial Appendage closure device? It’s hard to see the agency going against the 13:1 vote in favour from its expert panel.
Shrinking Pacemakers: First Human Implant of World’s Smallest Yet
Medtronic, Inc. is trumpeting the first-in-human implant of the world’s smallest pacemaker, called the Micra™ Transcatheter Pacing System. The device was implanted in a patient in Linz, Austria as part of the Medtronic global pivotal clinical trial.
Sorin Group Launches Intensia Family of ICD and CRT-D Devices Featuring DF-4 High Voltage Connector
Italian Cardiovascular device maker Sorin Group has gained CE Mark approval for its INTENSIA family of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy (CRT-D) devices, all of which feature a DF-4 high voltage connector.
Internal Fault Prompts FDA Safety Communication on Philips’ HeartStart Auto Defibrillators
Certain HeartStart automated external defibrillator (AED) devices made by Philips Medical Systems, a division of Philips Healthcare, may be unable to deliver needed defibrillator shock in a cardiac emergency situation, the US FDA has said in a new safety communication for users of these previously recalled devices.
FDA Approves Trial Expansion of Biotronik’s MRI Compatible Pacemakers
Biotronik, a pioneer in MRI compatible Cardiovascular implants devices, tells us that the US FDA has approved an expansion of its ongoing ProMRI® trial. This will allow inclusion of additional patient cohorts into the study, providing further support for the company’s proposed claims.
CE Mark and First Implant of Sorin’s KORA 100 MRI-Compatible Pacing System
Italian Cardiovascular device company Sorin has confirmed that it has gained CE Mark approval and seen a first implant of the KORA 100 pacing system, which is MRI compatible.
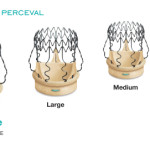
CE Mark for Extra Large Sizes of Sorin’s Perceval™ Sutureless Aortic Valve
Last week we covered the news that Italian cardiovascular outfit Sorin had gained CE mark approval for its Solo Smart stentless heart valve. Now the company tells us it has also gained the European nod for the large sizes of its sutureless Perceval valve.
FDA Approval for Boston’s Promus Premier™ “Next Generation” Drug Stent
Boston Scientific has followed its July CE mark approval of the Promus Premier drug eluting stent, with a newly announced FDA approval. The company claims “next generation” status, based on enhancements in stent design and delivery system.
CE Mark and First Implant for Sorin’s “Removable Stent” Biological Valve
Sorin gets CE mark approval for Solo Smart,the evolution of Freedom Solo, a stentless and biological aortic valve with no synthetic material, proven outstanding hemodynamic and clinical performance.
Boston Scientific’s Lotus™ TAVI Sees First Commercial Implants in Europe
Last month we noted the CE mark approval of Boston Scientific’s Lotus™ transcatheter heart valve, which was indicated for use in aortic valve patients for whom conventional surgery was a high risk option. Now the first two commercial implants of the Lotus Valve have taken place in a German hospital.
Delamination Risk Prompts FDA Class I Recall of Medtronic’s Guidewires
The US FDA has issued its highest category of of recall (Class I) for a variety of Medtronic Guidewires, due to the potential for their PTFE coating to part company with the core.
Successful Transendocardial Delivery of Autologous Cells for Ischemic Heart Failure
It’s American Heart Association(AHA) meeting week in Dallas, Texas, and cardiovascular regenerative medicine company BioCardia®, Inc. has updated us on trial results from its stem cell studies, as reported both at the congress and concurrently the Journal of the American Medical Association.
CoreValve® Back in the Game with German Court Appeal Win
Medtronic, Inc. has announced that a German court has ordered the discontinuation, in its entirety, of a prior court ruling that prohibited the company from commercially marketing or selling the CoreValve® System in Germany since August 26, 2013.
CE Mark for ReliantHeart HeartAssist5® Ventricular Assist Device
The ReliantHeart HeartAssist 5®Ventricular Assist Device (VAD) has gained CE Mark approval in Europe for use in patients requiring ventricular support due to end stage heart failure. The HeartAssist 5® is approved for bridge to transplant (BTT) patients awaiting cardiac transplantation and for destination therapy (DT) patients who are not candidates for cardiac transplantation.
All Eyes on Watchman FDA Panel Meeting in December
December 11th sees the FDA’s Circulatory System Devices Panel meet to discuss, make recommendations, and vote on information related to the premarket approval application regarding the Boston Scientific WATCHMAN Left Atrial Appendage Closure Technology.