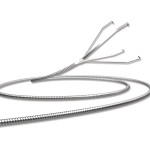
Covidien plc has unveiled an advanced ablation system that offers physicians predictable results regardless of the target location or tissue type.
Covidien
Covidien Launches Enhanced Trellis Clot-Busting System
Covidien’s Trellis Peripheral infusion system features enhanced drug delivery and better clot removal.
European Launch of Covidien’s Pipeline™ Flex Embolization Device
Covidien has announced the launch of its new Pipeline Flex Neuro-Embolization device which is a minimally-invasive flow diversion device for unruptured brain aneurysms, now claiming more accurate, controlled placement.
Tomorrow Your Company’s Ours: Medtronic Swallows Covidien
With a combined revenue of $27 Billion, the acquisition of Covidien by Medtronic makes for one helluva company that promises to have fingers in every conceivable medtech pie.
FDA Approval for Covidien’s Neurovascular Micro Catheter
Covidien’s Apollo™ Onyx™ delivery Micro Catheter with Detachable Tip has gained FDA approval and will enhances the company’s Onyx™ Liquid Embolic System.
CE Mark and EU Launch of Covidien’s Nellcor™ Respiration Rate Technology
Covidien plc, has announced the European commercial launch of its Nellcor™ Respiration Rate System
Catch-up: Medtronic Wins CoreValve Stay and Covidien’s Pipeline Recall Gets FDA Class
Two of last week’s news items have moved on a bit over the Easter weekend: Medtronic’s increasingly acrimonious spat with Edwards over the respective companies’ transcatheter heart valve offerings, and Covidien’s recall being graded Class I by the FDA.
FDA Clears Disposable Feeding Tube Placement Under Direct Vision
Covidien plc has gained U.S. FDA 510(k) Clearance for its Kangaroo™ feeding tube with IRIS Technology. This first-of-its-kind camera-equipped disposable feeding tube is designed to enhance patient safety by providing visualization for a procedure that is currently blinded.
Delamination Risk Provokes Covidien Recall of PTFE Coated Vascular Products
Covidien plc has notified customers of a voluntary recall to address an issue with certain lots of its Pipeline™ Embolization Device and Alligator™ Retrieval Device where the polytetrafluoroethylene (PTFE) coating applied to the delivery wire could delaminate and detach from the devices.
Barrx Treatment Group Significantly Outperforms Control in Barrett’s Esophagus
Results of a new clinical study into the Barrx RF Ablation platform, acquired by Covidien back in late 2011, say the therapy is effective at treating Barrett’s esophagus. So effective that the trial was stopped and the control group offered the treatment.
FDA Clears Covidien’s Puritan Bennett™ 980 Ventilator
Covidien plc has announced its Puritan Bennett™ 980 ventilator has received U.S. FDA 510(k) clearance. The company says its new acute care ventilator is designed to be simple, safe and smart and to enable patients to breathe more naturally than with conventional mechanical ventilators.
Covidien Can Start Pillcam® Sales Now Given Imaging Deal is Done
Two months after announcing the agreement to acquire Given Imaging Ltd., Covidien now tells us it has completed the deal, which means the medtech giant now has a highly complementary range of swallowable endoscopic cameras in its bag.
FDA Clearance for Covidien LigaSure™ Maryland Jaw
Covidien has received U.S. FDA 510(k) clearance for the LigaSure™ Maryland jaw open/laparoscopic sealer/divider. In addition the company has completed all European requirements to CE Mark the product. It expects to launch the new vessel sealer and divider in the United States and the European Union during the current quarter.
Has Covidien Realised the Emperor’s Naked?
Is Covidien’s sudden exit from the erstwhile next big thing that was Renal Denervation therapy a sign that they’re not up for the fight? Or is a combination of slow adoption and a clinical mountain to climb making this massive market opportunity shrink before their eyes to the extent that it’s no longer viable?
It’s Win-Win: Confluent Suit Fits Integra Better than it did Covidien
Covidien has completed the sale of its Confluent Surgical product line to Integra LifeSciences Corporation for approximately $235 million in cash and is seemingly pleased to be rid of it. Integra will benefit from a highly complementary product.
Covidien Swallows Up Pillcam® Technology with Given Imaging Acquisition
Covidien plc is to acquire Israeli Pillcam® purveyor Given Imaging, Ltd. in a deal worth $860 million net of cash and investments.