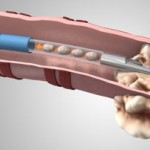
As it strives to gain U.S. approval, this most innovative of transcatheter heart valve developers has enlisted the services of an industry regulatory veteran
Products
Only MR-Conditional Rechargeable Spinal Cord Stimulator With Burst Stimulation Gets CE Mark
St. Jude has gained CE mark approval for its Prodigy MRI™ spinal cord stimulation therapy, the first of its kind to offer MRI compatibility and Burst stimulation
FDA 510(k) Clearance for Zyga SImmetry® Sacroiliac Joint Fusion System Updates
New clearance extends system’s indications to include new MIS approaches for Sacroiliac Joint fusion
Enrollment Complete in HeartWare’s Endurance2 HVAD Trial
Device aims to be indicated as a destination therapy rather than as a bridge to transplantation
Packaging Particulates Provoke Recall of Medtronic TAVI Loading System
Particulate matter in the delivery system of Medtronic’s newly FDA approved CoreValve Evolut R system present enough risk to warrant and FDA Class 1 Recall
FDA Approval of Apollo Endosurgery ORBERA™ Intragastric Balloon
When it comes to weight loss, Orbera Balloon fills a gap
U.S. Next? Sorin Gains Health Canada Approval for Perceval Valve
Sorin’s Sutureless Valve is in the process of undergoing U.S. FDA approval, but in the meantime is beefing up its international presence with the go-ahed in another market
EU Launch of Sorin’s REPLY CRT-P, World’s Smallest Cardiac Resynchronization Pacemaker
Tiny device offers patient benefit of minimal size combined with functionality including sleep apnea monitoring
Codman Neuro Announces US Launch of New Brain Aneurysm Coils
Coil shapes for every step of procedure combine with new delivery system
FDA Clearance for Medrobotics® Flex® Robotic System
“I’ve been a surgeon since 1960 and this technology is one of the most significant operating room advances I’ve seen in decades”
First U.S. Patients for Biotronik’s Newly Approved Itrevia HF-T QP CRT-D Device
Device incorporates latest defibrillator technology, closed loop stimulation and compatibility with a quadripolar lead
Medtronic’s New Biopsy System Targets Improved Lung Cancer Diagnosis
Medtronic tells us its recently FDA cleared system can get to difficult-to-access areas and collect larger, more intact samples
Mitek Launches New VAPR® RF Arthroscopic Ablation Devices
Company claims a new level of performance, procedural efficiency and safety as devices promise better ablation combined with lower temperature
U.S. Launch for K2M’s MESA® 2 Deformity Spinal System
Company says the new system carries forward the hallmarks of its complex spine MESA technology, while offering new capabilities to enhance user experience.
CE Mark for SinuSys™ Vent-Os® Sinus Dilation System for Treatment of Frontal and Sphenoid Sinusitis
Two new devices will expand portfolio of low-pressure, self-expanding dilation devices to treat frontal and sphenoid sinusitis, in addition to maxillary disease.
Aurora Spine Launches MIS VOX™ Lateral Interbody Spinal System Featuring TiNANO® Coating
Spine device company Aurora Spine Corporation has launched another new product in its line of next generation MIS lumbar interbody cages.