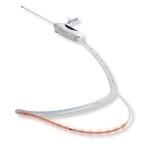
Breathe Technologies has gained FDA 510(k) clearance for its Life2000 Ventilation System for people needing mechanical ventilation
Products
New joimax iLESSYS Delta System for Treatment of Spinal Stenosis
joimax’s new spinal stenosis system claims to offer a new level of decompression with the option of cage implantation in parallel
Medtronic Launches GastriSail™ Bariatric Surgery Device
New Bariatric Surgery device makes the most common weight loss procedure in U.S. more consistent and efficient
Size Matters as Smith & Nephew Pulls Smaller Hip Resurfacing Components
Smaller sized Birmingham Hip Resurfacing components are being withdrawn due to heightened risk of revision surgery
CE Mark for St. Jude’s “Industry First” Neuromodulation Trial System
St. Jude Medical, Inc., has gained CE Mark approval and announced the European launch of its Invisible Trial System, an app-based and wireless neuromodulation programming system leveraging Apple™ iPod touch™ and iPad mini™ technology.
Direct Flow Medical® Names Co-principal Investigator for U.S. Pivotal Study
Transcatheter valve implantation using the novel Direct Flow “inflatable” valve has moved a step closer as the company appoints study leader
Coming Soon to an Abdomen Near You? TransEnterix Submits 510(k) Application for Surgical Robot
First patient-side robotically enhanced laparoscopic surgery platform aims for U.S. market clearance
Taking the Guesswork out of Epidural’s: FDA Clears Ultrasound Guidance System
Rivanna Medical’s Accuro™ system is the first ultrasound guidance system to provide 3D navigation to an anatomical target
DePuy Synthes Determinedly Integrates into Healthcare Provision
DePuy Synthes is launching its eSIMS Inventory Management and Ordering Solution as well as its Geriatric Fracture Program in the EMEA region
FDA Clears Penumbra’s ACE64 Thrombectomy System for Stroke Treatment
Latest mechanical thrombectomy device delivers higher revascularization rates, reduced procedure time and cost
New Decompressive Knee Brace from Townsend Design
It seems wearability is the order of the day as the company broadens patient demographic with new OA brace
CE Mark for Abbott’s New Absorb GT1™ Stent System
New generation of Abbott’s absorbable stent line features improved access and deliverability
FDA (510k) Clearance for Magstim’s rTMS (repetitive Transcranial Magnetic Stimulation) Therapy System
Pioneering device manufacturer cleared for U.S. launch of its Rapid2 Therapy System for treatment of drug resistant Major Depressive Disorder
CE Mark for Second Generation Endostim Neurostimulation Device for GERD
Endostim’s thinner, smaller new lower esophageal sphincter stimulating device promises an improved patient experience
CE Mark for Arterial Remodeling Technologies’ Pure Bioresorbable Scaffold
Arterial Remodeling Technologies has gained CE Mark approval for its next generation drug free, pure bioresorbable scaffold used to treat coronary artery disease
CE Mark of Perivascular Alcohol Denervation System for Hypertension
CE Mark of the Ablative Solutions Peregrine System™ Infusion Catheter signals continued interest in renal denervation therapy