MRI-conditionality is rapidly becoming industry standard, at least in St. Jude’s range, as more ICD products are approved to make this claim in Europe
Products
Australian Approval for GORE® EXCLUDER Iliac Branch Endoprosthesis
Gore’s newly approved device builds on its Excluder family of AAA treatment devices and is the first dedicated for use in the common iliac artery
FDA Approval and CE Mark for Medtronic’s Arctic Front Advance® ST Cryoballoon
Based on clinician feedback Medtronic’s third generation cryoballoon nudges ablative treatment of atrial fibrillation along
FDA Green Light for StimGuard’s IDE Study into Implantable Overactive Bladder Device
Could StimGuard’s tiny implant ultimately eliminate expensive pulse generators?
Another Spinal Cord Stimulation System Gets Pain Indication
Nevro Corporation has received approval from the U.S. FDA for its Senza spinal cord stimulation (SCS) system for the treatment of chronic pain.
FDA Cleared for U.S. Launch: Medtronic’s Euphora™ Semicompliant Coronary Balloon
Following on the heels of its non compliant forebear, the Euphora semicompliant coronary balloon offers enhanced pushability
First Biosynthesized Cellulose-Based Dural Substitute Launched
Can DePuy Synthes CMF depose current incumbent dural subs with its new offering? Study results suggest it just might.
US Launch for Medicrea’s Second Generation LigaPass®
New improved spinal fixation connector now cleared for use in young and smaller patients
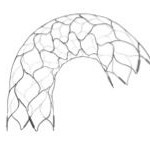
Early CE Mark for STENTYS’ Next-Generation Self-Apposing Stent System
“Quantum Leap” in design means Self-Apposing stent delivery now mimics conventional stent placement, says the company
New Combined Monitoring/Stabilisation Technology Offers Hope for Sleep Apnea Patients
Resmed says its AirCurve 10 CS PaceWave™ reinvents the management of Central Sleep Apnoea and could play a pivotal role in the care of millions of patients with concurrent chronic heart failure
CE Mark for BONESUPPORT CERAMENT™|V, Injectable Vancomycin-Eluting Bone Substitute
First Injectable Vancomycin Eluting Bone Substitute in the Management of Osteomyelitis gains regulatory approval in Europe
Codman’s New Intracranial Aneurysm Stent Boasts Greater Conformability, Visibility and Ease of Use
J&J company Codman Neuro has announced the launch of its new neurovascular stent in Europe.
Promise of Affordable Advanced Radiotherapy with Varian’s VitalBeam™
Varian Medical Systems has unveiled its VitalBeam™ system, which is being touted as a new cost-effective technology package for offering high-quality, high-throughput radiation therapy
First-of-its-Kind Approval for Near Vision Corneal Implant
The U.S. FDA has approved the first implantable device for correction of near vision in patients who have not had cataract surgery
Enova Illumination Launches World’s “Newest and Brightest” LED Surgical Headlight
Enova Illumination is celebrating its tenth anniversary and launching a new LED surgical headlight at the same time.
U.S. FDA Clearance for Two Pigmented Lesion Treatments
Two new clearances have been announced for technologies to treat a variety of aesthetic treatments