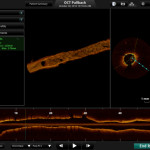
TCT2013 will see St. Jude Medical, Inc. showcasing its newly FDA approved ILUMIEN™ OPTIS™ PCI Optimization™ System, a new technology designed to provide physicians with a comprehensive disease assessment tool for treating patients with coronary artery disease.
Products
FDA 510(k) Clearance for Varian’s RapidPlan™ Brings Knowledge-Based Treatment Planning to Radiation Oncology
Radiotherapy expert Varian Medical Systems has received 510(k) clearance from the U.S. FDA for a radiotherapy treatment planning tool designed to enhance quality, consistency, and efficiency in radiotherapy treatment planning.
L5/S1 Lateral Disc Access with New OLIF51 System
Medtronic, Inc. has used the annual meeting of the Congress of Neurological Surgeons in San Francisco, at which to announce the launch of the first procedure that allows lateral access to the most common operative level of the spine.
Medtronic’s US Launch of Export Advance Coronary Aspiration Catheter
Medtronic, Inc. has announced the U.S. launch of its Export Advance aspiration catheter, which it says offers greater power and superior deliverability in coronary artery thrombus removal.
Deployment Problems Lead to FDA Recall for Bard’s LifeStent Solo Vascular Stent
The FDA has issued a Class I recall notice on certain product codes and lot numbers of Bard’s LifeStent Solo Vascular Stent, manufactured between November 2011 and June 2012. The reason for the recall is a potential failure to deploy the stent, which would have potentially serious adverse health consequences
Is There a Downside to Being a Guinea Pig?
Nothing’s perfect, so why regulate it as though it is, goes the argument against tighter regulations. Especially when doing so would mean less innovation, less investment and no obvious reason why patient safety would improve.
Serious Tissue Erosion Warning for St.Jude Amplatzer Atrial Septal Occluder
The FDA has issued an alert to health care providers and patients concerning its Amplatzer Atrial Septal Occluder (ASO). It seems that in very rare instances, tissue surrounding the device can erode, resulting in life-threatening emergencies that require immediate surgery.
CE Mark for Pulsar Vascular’s PulseRider®
Neurovascular technology company Pulsar Vascular has received European CE Mark approval for its lead product, the PulseRider®. The company says its device is designed to treat brain aneurysms in conjunction with embolic coils or flow diversion devices.
FDA Warns Globus over MicroFuse® Putty Deficiencies
It’s a bit of a “so-what” story really, but when the FDA sends you a warning that you haven’t acted on its observations it is kinda newsworthy. Globus says it’s on top of the situation.
LuViva Still Not There with FDA, Beefs Up International Effort
Guided Therapeutics has still not addressed all of the FDA’s concerns as it attempts to gain PMA approval for the LuViva cervical scanning device. It’s consoling itself with revenue from a newly enhanced push into more international markets.
FDA Clearance for Crospon EsoFLIP® Dilation Catheter
Irish MIS device company Crospon tells us it has received US FDA clearance for its EsoFLIP® balloon dilation catheter. The device is complementary to the Crospon’s EndoFLIP® measurement device and represents the company’s first foray into endoscopic therapeutic products.
Newly Cleared Integra® Hollywood™ NanoMetalene™ Interbody Device Brings Best of Both Worlds
Ingeniously, the newly FDA cleared Hollywood Nanometalene IBD from Integra comprises a PEEK-OPTIMA core construct, coated with an ultrathin layer of titanium, providing the best of both worlds in Interbody Device design.
Depuy Boss Worries About FDA and Sales Taxes. But EU Regs Changes Will Hit Just as Hard
When the boss of a big dog medtech company stands up to speak we all listen. And when he says a combination of FDA rules and medical device sales tax is hurting US industry we cannot disagree. But are these people ignoring the threat of newly restrictive EU regulations?
NASS Launch for Invuity® BREITEN™ Illuminated Retractor Offers “Unsurpassed” Anterior Cervical Disc Space Visualization
At this week’s North American Spine Society (NASS) Annual Meeting the company has introduced its Breiten™ Illuminated Retractor System. The system promises to increase surgical efficiency, reduce operating room time and lessen surgeon fatigue by eliminating or reducing dependence on a headlight
AAOMS 2013 Sees Depuy Synthes CMF Launch Integrated, Customized Facial Reconstruction
TRUMATCH® CMF Solutions Spans Preoperative
Virtual Surgical Planning Services, Intraoperative Tools and Customized Implants
K2M Launches SANTORINI® Small Corpectomy Cage at NASS
It’s 18 months since minimally invasive spine technology company K2M, Inc., gained FDA clearance for its Santorini® Corpectomy cage system. … continue reading “K2M Launches SANTORINI® Small Corpectomy Cage at NASS”