Boston Scientific Corporation has received FDA approval for its latest generation of defibrillators and heart failure devices, including the DYNAGEN™ MINI and INOGEN™ MINI ICDs, as well as the DYNAGEN™ X4 and INOGEN™ X4 CRT-Ds.
Regulatory
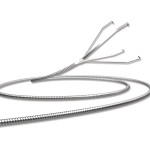
FDA IDE Approval for BARREL® Intracranial Bifurcation Aneurysm Solution
Reverse Medical® Corporation has gained FDA IDE approval for clinical investigation of its BARREL® Vascular Reconstruction Device (VRD). The BARREL® Vascular Reconstruction Device is designed for use with occlusive devices in the treatment of intracranial bifurcation aneurysms.
FDA Clears Disposable Feeding Tube Placement Under Direct Vision
Covidien plc has gained U.S. FDA 510(k) Clearance for its Kangaroo™ feeding tube with IRIS Technology. This first-of-its-kind camera-equipped disposable feeding tube is designed to enhance patient safety by providing visualization for a procedure that is currently blinded.
Delamination Risk Provokes Covidien Recall of PTFE Coated Vascular Products
Covidien plc has notified customers of a voluntary recall to address an issue with certain lots of its Pipeline™ Embolization Device and Alligator™ Retrieval Device where the polytetrafluoroethylene (PTFE) coating applied to the delivery wire could delaminate and detach from the devices.
FDA Backs Up Cochlear™Nucleus® Hybrid™Approval
Last month implantable hearing solutions expert, Cochlear Ltd., gained U.S. FDA approval for its Cochlear™ Nucleus® Hybrid Implant System. Now the FDA has issued its own information summary about the device.
CE Mark for Advanced Cooling Therapy’s Novel Esophageal Cooling Device
An emerging medical device manufacturing company (that we’d never come across until now), Advanced Cooling Therapy (ACT), has received CE marking for its first product, the Esophageal Cooling Device (ECD) to control patient temperature. The product is now available to medical professionals in Europe.
FDA Clears Ortho Development’s Ovation Tribute™ Hip Stem
Ortho Development® Corporation has received FDA clearance for its Ovation Tribute™ Hip Stem. The Ovation Tribute is a femoral hip prosthesis that is intended for cementless use in total hip replacement and hemiarthroplasty procedures.
Medtronic ICD System CE Marked for Full Body MRI Scans
Medtronic has gained CE Mark approval for its Evera MRI™ SureScan® implantable cardioverter-defibrillator (ICD) System. The company says this is the first and only ICD system approved for magnetic resonance imaging (MRI) scans positioned on any region of the body.
CE Mark for Abiomed Impella® RP
Abiomed has received CE Mark approval to market its Impella RP device in the European Union, providing a non surgical heart support option for patients with reduced right ventricular function.
FDA and CE Nods for Boston’s Expect™ Slimline (SL) Needle for Fine Needle Aspiration During Endoscopic Ultrasound
Boston Scientific Corporation has received U.S. FDA clearance and CE Mark approval in Europe for the Expect™ Slimline (SL) Needle, which means it is now globally available for Endoscopic Ultrasound-Fine Needle Aspiration (EUS-FNA) procedures.
CE Mark for EDWARDS INTUITY Elite Valve System Means Small-Incision Surgical Valve Replacement
Cardiovascular device specialist, Edwards Lifesciences Corporation has received CE Mark approval for its EDWARDS INTUITY Elite valve system. The company says its next-generation, rapid deployment system facilitates smaller incisions in surgical aortic valve replacement procedures
FDA Clears FasTouch™ Suture-Like Hernia Mesh Fixation System
Clever little device this, a thing looking like a glorified cable-tie, but deliverable laparoscopically using a 5mm diameter instrument, all aimed at fixing hernia mesh in place securely and with minimal foreign body.
St. Jude Claims “World First” as FDA Approves Upgradeable Spinal Cord Stimulator
St. Jude Medical, Inc., is trumpeting the U.S. FDA approval of its Protégé™ IPG, the first and only neurostimulation system that allows spinal cord stimulation (SCS) technology upgrades to be made via software updates.
First Patients Implanted with ICDs in BIOTRONIK ProMRI® Study
Cardiovascular device company Biotronik has announced that the first patients across the United States have received implants of the new Iforia implantable cardioverter-defibrillators (ICDs)
Euro Parliament Sets Regs Changes inTrain
The European Parliament is edging towards completion of the long-awaited steps that will spell a new regime for regulating medical devices in Europe.
FDA Clears Extremity Medical’s HammerFiX™ IP Fusion Device
Extremity Medical, LLC has received U.S. FDA 510(k) clearance for the HammerFiX™ for the fixation of osteotomies and reconstruction of the lesser toes following correction procedures for hammertoe, claw toe, and mallet toe.