C.R. Bard has announced the submission of the final module to support FDA approval for the Lutonix® Drug-Coated PTA Dilatation Balloon/Catheter. This could put it at the front of the queue of companies hoping for a first peripheral drug coated balloon approval.
Regulatory
CE Mark for eZono 4000 Tablet Ultrasound System with eZGuide
German “tablet” ultrasound specialist eZono AG, has received CE marking for its eZono 4000 system, with eZGuide, a revolutionary needle guidance technology.
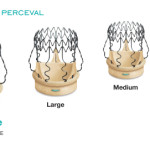
CE Mark for Extra Large Sizes of Sorin’s Perceval™ Sutureless Aortic Valve
Last week we covered the news that Italian cardiovascular outfit Sorin had gained CE mark approval for its Solo Smart stentless heart valve. Now the company tells us it has also gained the European nod for the large sizes of its sutureless Perceval valve.
FDA Approval for Boston’s Promus Premier™ “Next Generation” Drug Stent
Boston Scientific has followed its July CE mark approval of the Promus Premier drug eluting stent, with a newly announced FDA approval. The company claims “next generation” status, based on enhancements in stent design and delivery system.
KCI Joins Portable NPWT Race with Next Generation V.A.C.Via™ Therapy System
The US FDA has cleared V.A.C.®Via™ Negative Pressure Wound Therapy System from Kinetic Concepts, Inc., featuring a diaphragm pump for faster draw down, longer battery life and higher leak rate threshold than its forebear.
CE Mark and First Implant for Sorin’s “Removable Stent” Biological Valve
Sorin gets CE mark approval for Solo Smart,the evolution of Freedom Solo, a stentless and biological aortic valve with no synthetic material, proven outstanding hemodynamic and clinical performance.
Tensyn™ Band 510(k) For Syndesmosis Repair: Come on Dallen, Show Us Your Wares
Back in July, Dallen Medical submitted its 510(k) for the Tensyn™ band for fixation of ankle syndesmosis. Now the company is reporting that the FDA has duly cleared the product for market.
Final PMA Module Submitted for TissuGlu® Surgical Adhesive
Surgical Adhesive manufacturer Cohera Medical, Inc.® has submitted its fourth and final FDA PMA module that would ultimately see TissuGlu® indicated for reducing drainage following abdominoplasty procedures in the US.
US and EU Nods for Boston Scientific’s Direxion™ Torqueable Microcatheter
Boston Scientific’s FDA cleared and CE mark approved new embolectomy catheter features a proprietary shaft design that the company says enables better control In hard-to-navigate Vessels.
Delamination Risk Prompts FDA Class I Recall of Medtronic’s Guidewires
The US FDA has issued its highest category of of recall (Class I) for a variety of Medtronic Guidewires, due to the potential for their PTFE coating to part company with the core.
FDA Approval Brings New Treatment Option for Uncontrolled Epilepsy
Neuro implant developer, NeuroPace has announced the U.S. FDA approval of its RNS Stimulator, a device to help reduce the frequency of seizures in epilepsy patients who have not responded well to medications.
Newly CE Marked Tissue Damage Detector Launched at Medica
Bruin Biometrics, LLC has gained CE Mark approval to market the SEM Scanner in the European Union. The SEM Scanner is that is intended to detect localized tissue edema and pressure induced tissue damage that may lead to the formation of pressure ulcers before damage becomes visible on the skin’s surface.
CE Mark for ReliantHeart HeartAssist5® Ventricular Assist Device
The ReliantHeart HeartAssist 5®Ventricular Assist Device (VAD) has gained CE Mark approval in Europe for use in patients requiring ventricular support due to end stage heart failure. The HeartAssist 5® is approved for bridge to transplant (BTT) patients awaiting cardiac transplantation and for destination therapy (DT) patients who are not candidates for cardiac transplantation.
All Eyes on Watchman FDA Panel Meeting in December
December 11th sees the FDA’s Circulatory System Devices Panel meet to discuss, make recommendations, and vote on information related to the premarket approval application regarding the Boston Scientific WATCHMAN Left Atrial Appendage Closure Technology.
CE Mark for Avedro’s KXL II ™ Non-Surgical Myopia Correction System
Avedro Inc., a Boston-based ophthalmic medical device and pharmaceutical company, has gained CE Mark clearance for its KXL II System, building on the company’s portfolio of technologies aimed at non-surgical correction of ophthalmic conditions.
Medtronic’s Software Gains FDA Nod for Use in Detecting ICD Lead Issues with Non-Medtronic Leads
Medtronic’s Lead Integrity Alert software is designed to detect implantable pacing lead failures better than impedance monitoring alone. Now the company has gained FDA approval for its system to be used in the detection of problems with other manufacturers’ leads, which opens up a further 12,000 case in the US alone.