Another storm-weathering exercise sees GI Dynamics reverse-splitting its stock on a 1:10 basis as it attempts to shore up its ailing share price
General Surgery
New Data Supports Exalenz’s BreathID® Test for Detecting H. Pylori in Emergency Setting
Newly published data suggest a Test-and-Treat approach to H. Pylori infection is viable and may carry several health benefits
Boston Scientific Targets Endoscopic Ultrasound Leadership with Xlumena Buy
Boston Scientific Corporation is to acquire Xlumena, Inc., developer of Endoscopic Ultrasound-guided transluminal drainage devices
AMA Publishes Category I CPT Code for Trans-Orifice Esophagogastric Fundoplasty
Code represents important first step In process to gain coverage for incisionless procedures to treat the underlying cause of GERD
EndoBarrier® Trial Halted and Stock Suspended
Followers of the EndoBarrier story will be wondering what happens next as the product’s maker is hit by more bad news
Cash Injection Means Gecko Biomedical Can Stick to its Plan
They might have moved on from trying to mimic the lizard, but nonetheless this French company has surgical adhesive aspirations that will be facilitated by this cash injection.
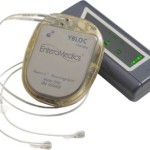
FDA Approves First Obesity Treatment Device for Ten Years
Enteromedics is celebrating approval of its novel neuroscience-based technology that it says enables safe, durable eeight loss
Covidien’s PILLCAM® SB Capsule Endoscope Featured in New Smithsonian Exhibit
Innovative design of wireless, vitamin-sized capsule recognized for its profound impact on improving the standard-of-care for small bowel examinations
Bowel Glue Jumps Queue
Cohera Medical, Inc. has received Priority Review Status Designation from the US FDA for its Sylys® Surgical Sealant
NICE Consults on Non-Surgical Device for Liver Diagnosis
UK NHS Watchdog NICE is consulting on draft guidance on a device that avoids surgical biopsy as it aims to diagnose and monitor liver damage.
Staff Cuts as GI Dynamics “Re-Evaluates Corporate Strategy”
It doesn’t sound like GI Dynamics is in a good place at the moment as it loses staff in an effort to retrench.
Cohera’s Hopes for GI Anastomosis Trial of Sylys® Surgical Sealant
Proving efficacy of a surgical sealant in GI anastomosis is likely to be difficult, but Cohera hopes its Sylys IDE will hit the spot.
FDA Clears Aer-O-Scope™ 360° Colonoscope System
GI View Ltd., has announced that it has received U.S. FDA 510(k) clearance for its flagship product, the Aer-O-Scope™ Colonoscope System
EndoBarrier® Back on EU Market, with Revised IFU
It seems the suspension of EndoBarrier sales in Europe it was more than just a paper exercise, as vigilance changes have seen it change its indications.
Cardica Calls Time-Out to Re-Think MicroCutter XCHANGE 30®
Cardica, Inc. is to pull the remainder of the latest iteration of its MicroCutter XCHANGE 30® from sale, drawing a line under the device until a next generation version is ready.
Promise of Less Tissue Slippage with Ethicon’s New Echelon Flex™ GST Powered Stapler
Reducing tissue slippage is a desirable feature of any laparoscopic cutting device, delivering more consistent results and potentially fewer firings.