When the boss of a big dog medtech company stands up to speak we all listen. And when he says a combination of FDA rules and medical device sales tax is hurting US industry we cannot disagree. But are these people ignoring the threat of newly restrictive EU regulations?
DePuy
AAOMS 2013 Sees Depuy Synthes CMF Launch Integrated, Customized Facial Reconstruction
TRUMATCH® CMF Solutions Spans Preoperative
Virtual Surgical Planning Services, Intraoperative Tools and Customized Implants
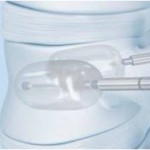
What’s New from Depuy Synthes Spine at NASS
Depuy Synthes Spine is launching its SYNFLATE Vertebral Balloon at NASS, claiming it offers a versatility not found in other vertebral body augmentation portfolios. At the same event the company is announcing the “mPACT” program targeting posterior mini open spinal fixation and fusion procedures.
MIS Rib Fracture Fixation with US Launch of Depuy Synthes CMF MatrixRIB™ System
DePuy Synthes CMF has announced the U.S. launch of new instrumentation that enables less invasive surgical fixation and stabilization of rib fractures. The MatrixRIB™ System offers precontoured, low-profile titanium plates, locking screws and intramedullary splints.
Mitek’s New Cuff Anchor Launched At AOSSM 2013
It’s sports med week this week, with the annual gathering that is the AOSSM meeting taking place in Chicago. Mitek Sports Medicine, a DePuy Synthes Company, under the wing of Johnson & Johnson, has used the event to showcase its new HEALIX ADVANCE™ KNOTLESS Anchor, a new suture anchor it says is designed to provide optimal knotless fixation for rotator cuff repair.
Launch For Mitek Sports Med’s New Small Diameter Dual-Thread Suture Anchor And New PTRC Repair System
Mitek Sports Medicine has unveiled the Healix 3.4mm Suture Anchor, its smallest dual-thread offering for rotator cuff repair. This coincides with the introduction of a new double-loaded minimally invasive partial thickness rotator cuff repair system, as part of its Healix Transtend™ Implant System.
FDA Urged To Require Safety Testing Of Metal-on-Metal Hips
Consumer protection activist organisation, Consumers Union has urged the U.S. FDA today to adopt a proposed rule requiring metal hip implants to be reviewed through the agency’s premarket approval (PMA) process to prove that the devices are safe and effective.
Wow! ASR Jury Finds “For J&J”
Now we weren’t expecting that. With DePuy ASR trials there was a thought that it would be a matter of how much rather than whether there was an award or not. Seemingly not true as an Illinois jury finds in favour of the company and offers no settlement.
Mitek Showcases New Biocomposite Interference Screw Design At AAOS
It’s AAOS week, and Mitek Sports Medicine has chosen the event for the launch of the MILAGRO® Advance Interference Screw, a new biocomposite screw designed to offer easier and faster insertion when compared to traditional interference screws.
The World vs J&J DePuy: Round 2
Case number 2. Only 10,748 to go. This time its Chicago and one Carol Strum vs DePuy. Early shots suggest they’re aiming at punitive damages based on the assertion that DePuy knew of the design flaws in its product before Ms Strum had one implanted in her.
Metal-on-Metal Vultures Circling
Law firm, Legal Bay has issued this release following the jury’s massive award to Loren Kransky, plaintiff against J&J DePuy. It’s predicting many many more such cases.
Unpicking The Kransky/DePuy Outcome
Loren Kransky has been awarded over $8M in his lawsuit against J&J Depuy over the ASR hip prosthesis. J&J won’t know whether to laugh or cry right now. At least Mr Kransky wasn’t awarded punitive damages, but future cases might challenge that finding again..and again.
Kransky vs J&J DePuy Trial Closing Remarks
Here’s an update on the court case currently underway in the U.S. which may prove a bellwether for future proceedings … continue reading “Kransky vs J&J DePuy Trial Closing Remarks”
Kransky vs DePuy: Chromium Didn’t Poison Patient Says Toxicologist
So did chromium from wear particles in DePuy’s ASR hip system poison Mr Kransky? A prominent toxicologist says no, but then his work is undermined by the plaintiff’s attorney who implies bias. It’s feisty stuff.
FDA Class I Recall As DePuy’s LPS Diaphyseal Sleeve May Not Accommodate Physiologic Loads
The FDA has issued a Class I recall of DePuy’s LPS Diaphyseal Sleeve the company having found that the Sleeve to its Base taper connection may not be sufficient to accommodate potential physiologic loads under some circumstances.
DePuy “did more testing on the ASR than any other product we put on the market”
Update on proceedings between “vasculopath” Loren Kransky and J&J (DePuy), centring on whether the company poisoned the plaintiff.