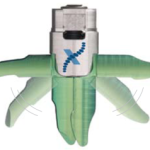
Boston Scientific Corporation has received U.S. FDA clearance and CE Mark approval in Europe for the Expect™ Slimline (SL) Needle, which means it is now globally available for Endoscopic Ultrasound-Fine Needle Aspiration (EUS-FNA) procedures.
Products
AANS sees Debut of DePuy’s ANSPACH® EG1 High Speed Drill System
DePuy Synthes Power Tools has launched its “Next Generation”, 30% more powerful electric drill system for neurosurgery, otology and spinal procedures.
CE Mark for EDWARDS INTUITY Elite Valve System Means Small-Incision Surgical Valve Replacement
Cardiovascular device specialist, Edwards Lifesciences Corporation has received CE Mark approval for its EDWARDS INTUITY Elite valve system. The company says its next-generation, rapid deployment system facilitates smaller incisions in surgical aortic valve replacement procedures
FDA Clears FasTouch™ Suture-Like Hernia Mesh Fixation System
Clever little device this, a thing looking like a glorified cable-tie, but deliverable laparoscopically using a 5mm diameter instrument, all aimed at fixing hernia mesh in place securely and with minimal foreign body.
St. Jude Claims “World First” as FDA Approves Upgradeable Spinal Cord Stimulator
St. Jude Medical, Inc., is trumpeting the U.S. FDA approval of its Protégé™ IPG, the first and only neurostimulation system that allows spinal cord stimulation (SCS) technology upgrades to be made via software updates.
First Patients Implanted with ICDs in BIOTRONIK ProMRI® Study
Cardiovascular device company Biotronik has announced that the first patients across the United States have received implants of the new Iforia implantable cardioverter-defibrillators (ICDs)
Euro Parliament Sets Regs Changes inTrain
The European Parliament is edging towards completion of the long-awaited steps that will spell a new regime for regulating medical devices in Europe.
FDA Clears Extremity Medical’s HammerFiX™ IP Fusion Device
Extremity Medical, LLC has received U.S. FDA 510(k) clearance for the HammerFiX™ for the fixation of osteotomies and reconstruction of the lesser toes following correction procedures for hammertoe, claw toe, and mallet toe.
CE Mark for Airo® Mobile Intraoperative CT
Mobius Imaging has gained CE Mark approval for its Airo Mobile Intraoperative CT System, distributed under an exclusive sales and service agreement with German company Brainlab.
Mazor Robotics to Launch New Renaissance® Brain Module at AANS
This weekend sees the American Association of Neurological Surgeons (AANS) annual meeting, starting April 7, 2014. Mazor Robotics is using the occasion to debut and officially launch the new Renaissance Guidance System brain module, which it says is FDA-cleared for both spine and brain surgery.
Final PMA Module Submitted to the FDA, for Superion® Interspinous Spacer System
Minimally invasive spine technologies company, VertiFlex®, Inc., has announced submission of the final module of the pre-market approval (PMA) application to the U.S. FDA to support approval for its Superion® Interspinous Spacer System.
FDA Approves U.S. Clinical Trial Start for CryoLife’s PerClot®
CryoLife, Inc. has received approval of its Investigational Device Exemption (IDE) for PerClot from the U.S. FDA. This approval allows CryoLife to begin its pivotal clinical trial to gain approval to commercialize PerClot in the U.S.
FDA Clearance for Ablative Solutions Peregrine System™ Infusion Catheter
Ablative Solutions, Inc., has received U.S. FDA 510(k) clearance for its Peregrine System™ Infusion Catheter. The system uses a unique, patent-pending technology to deliver diagnostic and therapeutic agents directly to the perivascular layer that surrounds blood vessels.
Sorin’s Perceval™ Sutureless Aortic Valve Now CE Marked for Under 65s
Italian Cardiovascular device specialist Sorin Group tells us its Perceval™ sutureless aortic valve has received CE mark approval for adult age indication allowing treatment of a wider spectrum of patients with aortic stenosis and/or steno-insufficiency.
FDA Clearance for Rotation Medical’s Implantable Collagen Scaffold for Treating Rotator Cuff Disease
Rotation Medical, Inc. tells us it has received U.S. FDA 510(k) clearance for its implantable collagen scaffold technology. The new technology is designed for use during arthroscopic and mini-open procedures to help support clinical management and protection of tendon injuries in which there has been no substantial loss of tendon tissue.
Intuitive Surgical Introduces “Technological Leap Forward” with New daVinci® Xi™ System
Robot surgery device specialist Intuitive Surgical, Inc., has announced the U.S. FDA clearance and introduction of the da Vinci Xi Surgical System, which it says represents a technological leap forward in replacing large-incision abdominal surgeries (open surgery) with a minimally invasive approach.