W. L. Gore & Associates has seen the FDA extend its indication for the GORE® VIABAHN® Endoprosthesis to include treatment of stenosis or thrombotic occlusion at the venous anastomosis of synthetic AV access grafts.
Products
Panel’s Yes Vote Means FDA Will Find it Hard to Say No to Boston’s WATCHMAN™ LAA Closure Device
Could the FDA be about to approve a permanently indwelling, percutaneously inserted Left Atrial Appendage closure device? It’s hard to see the agency going against the 13:1 vote in favour from its expert panel.
New FDA Approval Means Larger Target Vessels for Reverse Medical’s Peripheral Vascular Embolizer
Reverse Medical® Corporation has gained FDA 510(k) clearance to market its MVP®-5 Micro Vascular Plug system for peripheral vessel embolization, together with news of the first US clinical case.
Now TYRX Antibacterial Envelope FDA Cleared for Use with Spinal Cord Neuromodulators
TYRX, Inc. has received U.S. FDA clearance to expand marketing indications of its AIGISRx® N Antibacterial Envelope to include use with spinal cord neuromodulators.
CE Mark for AMS RetroArc™ Retropubic Female Stress Urinary Incontinence Sling
American Medical Systems® Inc. (AMS), has announced that its RetroArc™ Retropubic Sling System for treatment of female stress urinary incontinence (SUI) has gained CE Mark approval.
Sorin Group Launches Intensia Family of ICD and CRT-D Devices Featuring DF-4 High Voltage Connector
Italian Cardiovascular device maker Sorin Group has gained CE Mark approval for its INTENSIA family of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy (CRT-D) devices, all of which feature a DF-4 high voltage connector.
Internal Fault Prompts FDA Safety Communication on Philips’ HeartStart Auto Defibrillators
Certain HeartStart automated external defibrillator (AED) devices made by Philips Medical Systems, a division of Philips Healthcare, may be unable to deliver needed defibrillator shock in a cardiac emergency situation, the US FDA has said in a new safety communication for users of these previously recalled devices.
Class II Recall for Stalling Surgical Robot
Intuitive Surgical has written to customers worldwide, and in the US the FDA has issued a Class II recall notice which contains the instruction that users should contact the company to arrange an inspection visit and repair or replace daVinci robotic surgery device components as necessary to avoid a friction-induced stalling problem.
FDA Approves Trial Expansion of Biotronik’s MRI Compatible Pacemakers
Biotronik, a pioneer in MRI compatible Cardiovascular implants devices, tells us that the US FDA has approved an expansion of its ongoing ProMRI® trial. This will allow inclusion of additional patient cohorts into the study, providing further support for the company’s proposed claims.
British Breast Implant Maker Sizes Up French Market
A division of GC Aesthetics, Nagor is the only breast implant manufacturer based in Britain. And in a piece of elegant commercial symmetry it has just announced that it is now launching its range in France, no doubt hoping its quality message and reputation will score heavily in the aftermath of a certain other silicone breast manufacturer.
FDA Clears BME’s HammerLock® Intramedullary Fixation System Upgrades for Hammertoe Deformities
BioMedical Enterprises (BME), Inc. has announced the FDA clearance of an upgrade to the patent-pending HammerLock® Nitinol Intramedullary Fixation System.
CE Mark and First Implant of Sorin’s KORA 100 MRI-Compatible Pacing System
Italian Cardiovascular device company Sorin has confirmed that it has gained CE Mark approval and seen a first implant of the KORA 100 pacing system, which is MRI compatible.
Bard’s Lutonix Could Be First Drug Coated Balloon to US Market
C.R. Bard has announced the submission of the final module to support FDA approval for the Lutonix® Drug-Coated PTA Dilatation Balloon/Catheter. This could put it at the front of the queue of companies hoping for a first peripheral drug coated balloon approval.
CE Mark for eZono 4000 Tablet Ultrasound System with eZGuide
German “tablet” ultrasound specialist eZono AG, has received CE marking for its eZono 4000 system, with eZGuide, a revolutionary needle guidance technology.
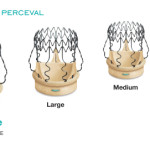
CE Mark for Extra Large Sizes of Sorin’s Perceval™ Sutureless Aortic Valve
Last week we covered the news that Italian cardiovascular outfit Sorin had gained CE mark approval for its Solo Smart stentless heart valve. Now the company tells us it has also gained the European nod for the large sizes of its sutureless Perceval valve.
FDA Approval for Boston’s Promus Premier™ “Next Generation” Drug Stent
Boston Scientific has followed its July CE mark approval of the Promus Premier drug eluting stent, with a newly announced FDA approval. The company claims “next generation” status, based on enhancements in stent design and delivery system.