Breakthrough technology offers fast, accurate, single-dose radiosurgery treatment planning for multiple metastases; helps to maintain patient comfort and spare normal tissue
Neuro
Codman Neuro Announces US Launch of New Brain Aneurysm Coils
Coil shapes for every step of procedure combine with new delivery system
FDA Clears Penumbra’s ACE64 Thrombectomy System for Stroke Treatment
Latest mechanical thrombectomy device delivers higher revascularization rates, reduced procedure time and cost
FDA (510k) Clearance for Magstim’s rTMS (repetitive Transcranial Magnetic Stimulation) Therapy System
Pioneering device manufacturer cleared for U.S. launch of its Rapid2 Therapy System for treatment of drug resistant Major Depressive Disorder
First Biosynthesized Cellulose-Based Dural Substitute Launched
Can DePuy Synthes CMF depose current incumbent dural subs with its new offering? Study results suggest it just might.
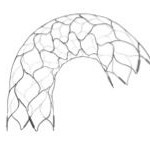
Codman’s New Intracranial Aneurysm Stent Boasts Greater Conformability, Visibility and Ease of Use
J&J company Codman Neuro has announced the launch of its new neurovascular stent in Europe.
Enova Illumination Launches World’s “Newest and Brightest” LED Surgical Headlight
Enova Illumination is celebrating its tenth anniversary and launching a new LED surgical headlight at the same time.
Orthofix Gains Option to Acquire eNeura and its Migraine Pain Treatment Device
eNeura’s SpringTMS® is the first such device to gain FDA clearance for migraine pain
FDA Approves Boston’s WATCHMAN™ Left Atrial Appendage Closure Device
First-Of-Its-Kind alternative to long-term Warfarin therapy for stroke risk reduction in patients with non-valvular atrial fibrillation
InVivo Woes Behind it as Clinicals and Board Changes Signal “Fruitful 2015”
2014 was a rollercoaster for the spinal scaffold developer, as the pilot study looked like it would take forever. That’s changed.
Ambitious Plans for Sorin/Cyberonics Following Merger
Sorin and Cyberonics Inc. are to merge, creating a new entity that they expect will be a major player in its combined fields
FDA Clears Office Alzheimers Test System
Neuronetrix’s Cognision™ system delivers the capability to test for Alzheimers in a primary care setting
Sequent Medical’s Latest Generation WEB Devices to be Included in U.S. IDE Study
The U.S. FDA has approved the inclusion of SL (“Single Layer”) family of WEB™ Aneurysm Embolization Device devices for use in its IDE study
Brain Disorder Diagnosis Needs Collaboration Among Neurological Researchers
Jan Medical is calling for global collaboration among neurological researchers to advance the science.
Medtronic Enjoys First Fruit of Covidien’s Product Pipeline
Medtronic has received U.S. FDA approval for the Pipeline™ Flex embolization device, recently absorbed into its product catalogue with the acquisition of Covidien.
Hope for Epilepsy Sufferers as NeuroPace RNS® System Reduces Seizures
Clinical study results suggest the NeuroPace RNS® System significantly reduces epileptic seizure frequency among certain adults