Regenerative medicine company Acell, Inc., has announced the launch of its new MatriStem® Multilayer Wound Matrix device for deep, hard-to-heal, full-thickness wounds.
Launches / Withdrawals
Zimmer Launches Puros® Demineralized Bone Matrix Block and Strip
Zimmer Spine’s flexible, cancellous bone allografts are used in spine surgery, either alone or as a bone graft extender to stimulate natural bone formation processes. New configurations make life easier for surgeons by reducing the need for shaping during procedures.
Brandon Medical’s Brilliant New LED Exam Light Solution
Leeds, UK-based Brandon Medical is launching a new range of LED examination lights that it claims are the most advanced of their type available anywhere in the World.
FDA Clearance for Covidien LigaSure™ Maryland Jaw
Covidien has received U.S. FDA 510(k) clearance for the LigaSure™ Maryland jaw open/laparoscopic sealer/divider. In addition the company has completed all European requirements to CE Mark the product. It expects to launch the new vessel sealer and divider in the United States and the European Union during the current quarter.
Europe-wide Release for Biotronik’s Passeo-18 Lux Peripheral Vascular Drug-Releasing Balloon
Cardiovascular device company Biotronik is in the news again with the announcement of its release of the Passeo-18 Lux Drug Releasing Balloon (DRB) in all countries accepting CE mark, following recent CE approval.
New Screw System Improves on K-Wires in Scapholunate Instability
Acumed has issued a press release trumpeting its new so-called SLIC Screw System for wrist reconstruction, which it says offers surgeons a new adjunct to soft tissue repair or reconstruction as it holds the bones in their reduced state while soft tissue repair heals.
U.S. Launch of Nuvasive’s ALIF ACR™
MIS spine device company NuVasive®, Inc. has announced the U.S. launch of its ALIF ACR™ (Anterior Lumbar Interbody Fusion – Anterior Column Realignment) system.
U.S. Launch for DePuy Synthes Trauma Modular Radial Head Prosthesis System
DePuy Synthes Trauma has announced the U.S. launch of a new Radial Head Prosthesis System for primary and revision joint replacement of the radial head to restore joint function.
Tissue Regenix Aiming High with US Distribution Plans for DermaPure™ Chronic Wound Therapy
UK wound healing expert company Tissue Regenix is planning to launch its biological scaffold product, DermaPure™, in early 2014 and is putting in place an extensive distribution network to make sure it gains appropriate coverage.
British Breast Implant Maker Sizes Up French Market
A division of GC Aesthetics, Nagor is the only breast implant manufacturer based in Britain. And in a piece of elegant commercial symmetry it has just announced that it is now launching its range in France, no doubt hoping its quality message and reputation will score heavily in the aftermath of a certain other silicone breast manufacturer.
Bigging Up the World’s Smallest CPAP machine
Human Design Medical, LLC is about to release the most versatile cpap device ever. The company says its Z1™ is a new innovative cpap device that is significantly lighter, smaller, quieter and more integrated than other cpap machines and is changing the rules of design, portability and user experience in the cpap industry.
Newly CE Marked Tissue Damage Detector Launched at Medica
Bruin Biometrics, LLC has gained CE Mark approval to market the SEM Scanner in the European Union. The SEM Scanner is that is intended to detect localized tissue edema and pressure induced tissue damage that may lead to the formation of pressure ulcers before damage becomes visible on the skin’s surface.
Paxeramed Debuts Industry’s First Social-media Medical Images Sharing Module
Medical imaging company Paxeramed has launched the industry’s first medical imaging sharing module to social media. The module anonymizes selected CT, MRI and X-Ray images and allows users to share it to social-media platforms from their reading workstations or dedicated viewing apps.
Platforms like Facebook and Twitter allow the shared images to be uploaded to private healthcare and hospital social media groups in a secure and HIPAA compliant manner.
First Patients for DJO’s Customised Shoulder System Planning Tool
Musculoskeletal device specialist DJO Global, Inc., tells us its Match Point System™ patient-specific shoulder instrumentation has seen its first use in Reverse® Shoulder System cases in Australia Florida.
New Entity SpineDirect’s Low Cost “No Rep” Strategy
Times are tough and healthcare providers are permanently squeezed. Does that mean a device company offering quality devices, but no rep support will be able to tap into the market on a cost basis?
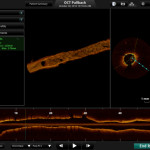
US Launch of St.Jude’s ILUMIEN™ OPTIS™ 3-D Vessel Reconstruction Technology
TCT2013 will see St. Jude Medical, Inc. showcasing its newly FDA approved ILUMIEN™ OPTIS™ PCI Optimization™ System, a new technology designed to provide physicians with a comprehensive disease assessment tool for treating patients with coronary artery disease.